3. Acids and Bases
Acid Base Equilibrium
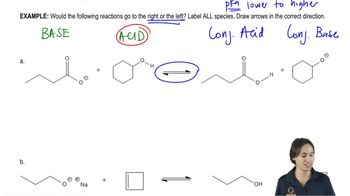
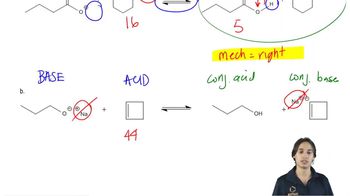
Problem 20b
Textbook Question
Textbook QuestionPhenols are less acidic than carboxylic acids, with values of pKa around 10. Phenols are deprotonated by (and therefore soluble in) solutions of sodium hydroxide but not by solutions of sodium bicarbonate. Explain how you would use extractions to isolate the three pure compounds from a mixture of p-cresol (p-methylphenol), cyclohexanone, and benzoic acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
999
views
Was this helpful?
Related Videos
Related Practice