24. Enolate Chemistry: Reactions at the Alpha-Carbon
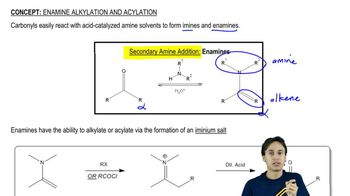
Enamine Alkylation and Acylation
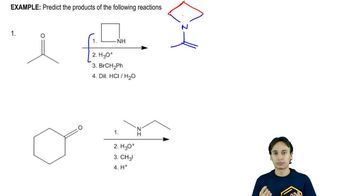
Problem 22.7
Textbook Question
Textbook QuestionWithout looking back, propose a mechanism for
the hydrolysis of this iminium salt to the alkylated ketone.
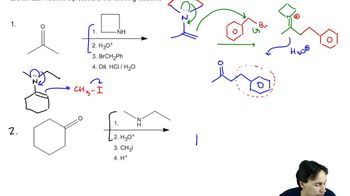
The first step is attack by water, followed by loss of a proton to give a carbinolamine. Protonation on nitrogen allows
pyrrolidine to leave, giving the protonated ketone.
<IMAGE>
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
78
views
Was this helpful?
Related Videos
Related Practice