3. Acids and Bases
Acid Base Equilibrium
Problem 2-13
Textbook Question
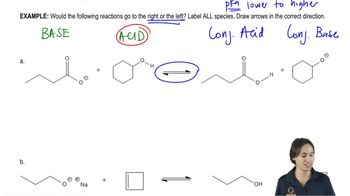
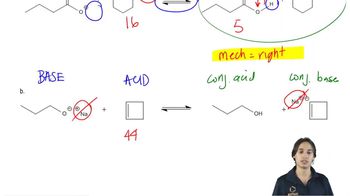
Textbook QuestionWrite equations for the following acid–base reactions. Label the conjugate acids and bases, and show any inductive stabilization. Predict whether the equilibrium favors the reactants or products. Try to do this without using a table of pKa values, but if you need a hint, you can consult [APPENDIX 4 <IMAGE]
g. NaOCH2CH3 + Cl2CHCH2OH
h. H2Se + NaNH2
i. CH3CHFCOOH + FCH2CH2COO-
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
107
views
Was this helpful?
Related Videos
Related Practice