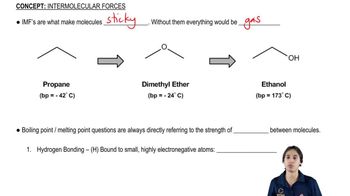
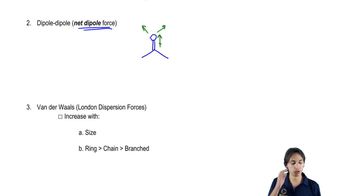
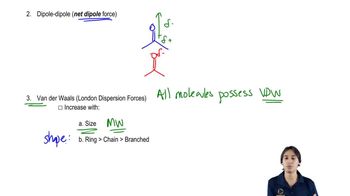
2. Molecular Representations
Intermolecular Forces
Problem 3.34a
Textbook Question
Textbook QuestionExplain why
a. H2O (100 °C) has a higher boiling point than CH3OH 165 °C2.
b. H2O (100 °C) has a higher boiling point than NH3 (-33 °C).
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
150
views
Was this helpful?
Related Videos
Related Practice