3. Acids and Bases
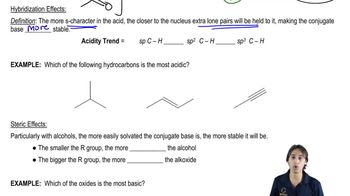
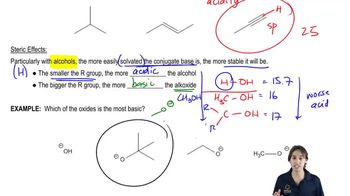
Ranking Acidity
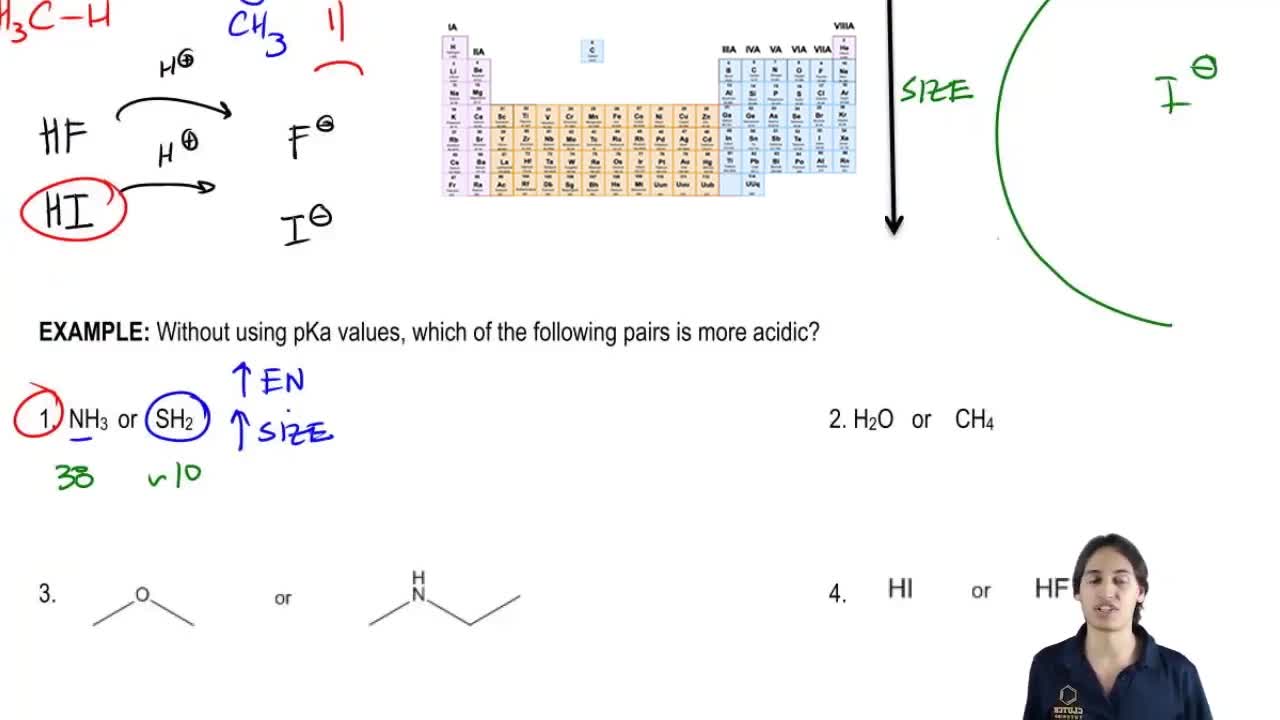
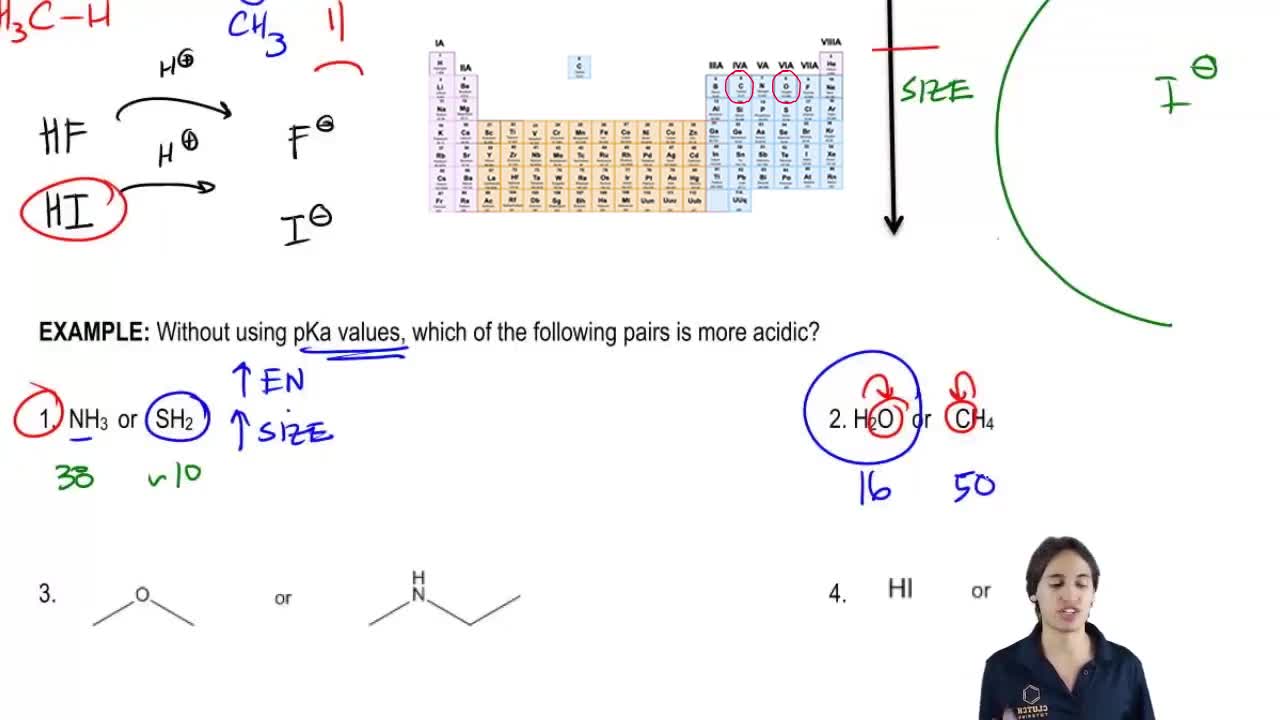
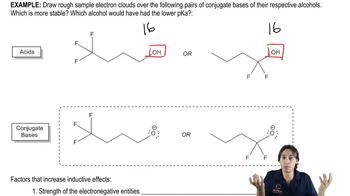
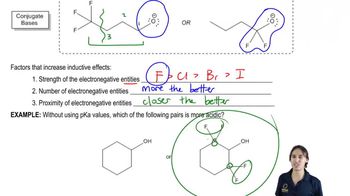
Problem 54
Textbook Question
Textbook QuestionThe pKa of ascorbic acid (vitamin C, page 55) <IMAGE> is 4.17, showing that it is slightly more acidic than acetic acid (CH3COOH, pKa 4.74).
a. Show the four different conjugate bases that would be formed by deprotonation of the four different OH groups in ascorbic acid.
b. Compare the stabilities of these four conjugate bases, and predict which OH group of ascorbic acid is the most acidic.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
112
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos