15. Analytical Techniques:IR, NMR, Mass Spect
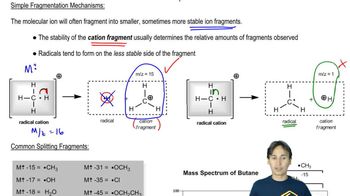
Mass Spect:Fragmentation
Problem 12e
Textbook Question
Textbook QuestionEthers are not easily differentiated by their infrared spectra, but they tend to form predictable fragments in the mass spectrum. The following compounds give similar but distinctive mass spectra.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
413
views
Was this helpful?
Related Videos
Related Practice