What are the answers to Problem 29 when the same compounds are treated with Br2 at 125 °C?
c.

 Verified step by step guidance
Verified step by step guidance
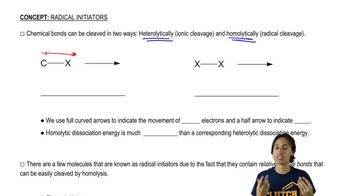
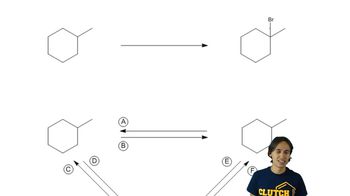

What are the answers to Problem 29 when the same compounds are treated with Br2 at 125 °C?
c.
What are the product(s) of each of the following reactions? Disregard stereoisomers.
f.
What are the product(s) of each of the following reactions? Disregard stereoisomers.
e.
What alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl2?
What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at room temperature? Disregard stereoisomers.
a.
What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at room temperature? Disregard stereoisomers.
c.