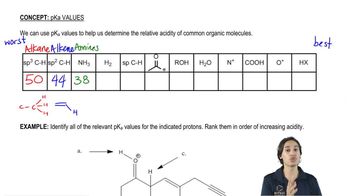
3. Acids and Bases
pKa
Problem 2.20
Textbook Question
Textbook QuestionAcetic acid can also react as a very weak base (pKb = 20). Two different sites on acetic acid might become protonated to give the conjugate acid.
1. Draw both of these possible conjugate acids, and explain (resonance) why the correct one is more stable.
2.Calculate the pKa of this conjugate acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
130
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos