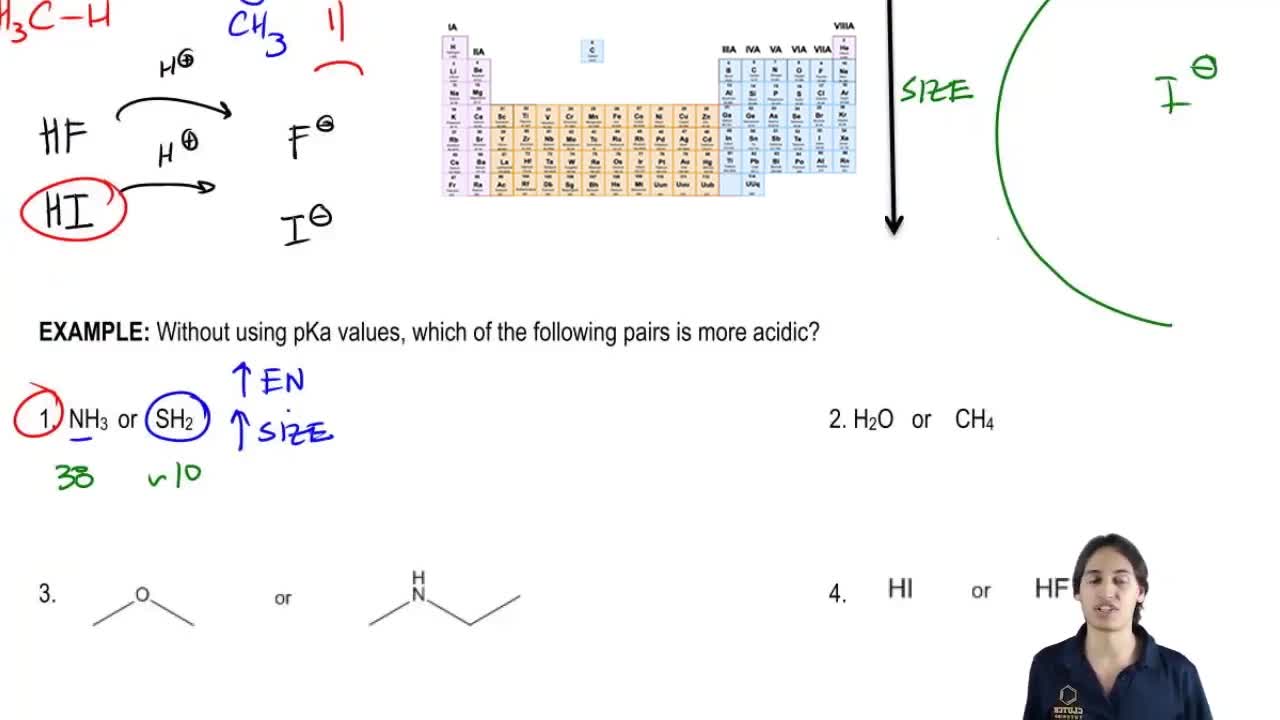
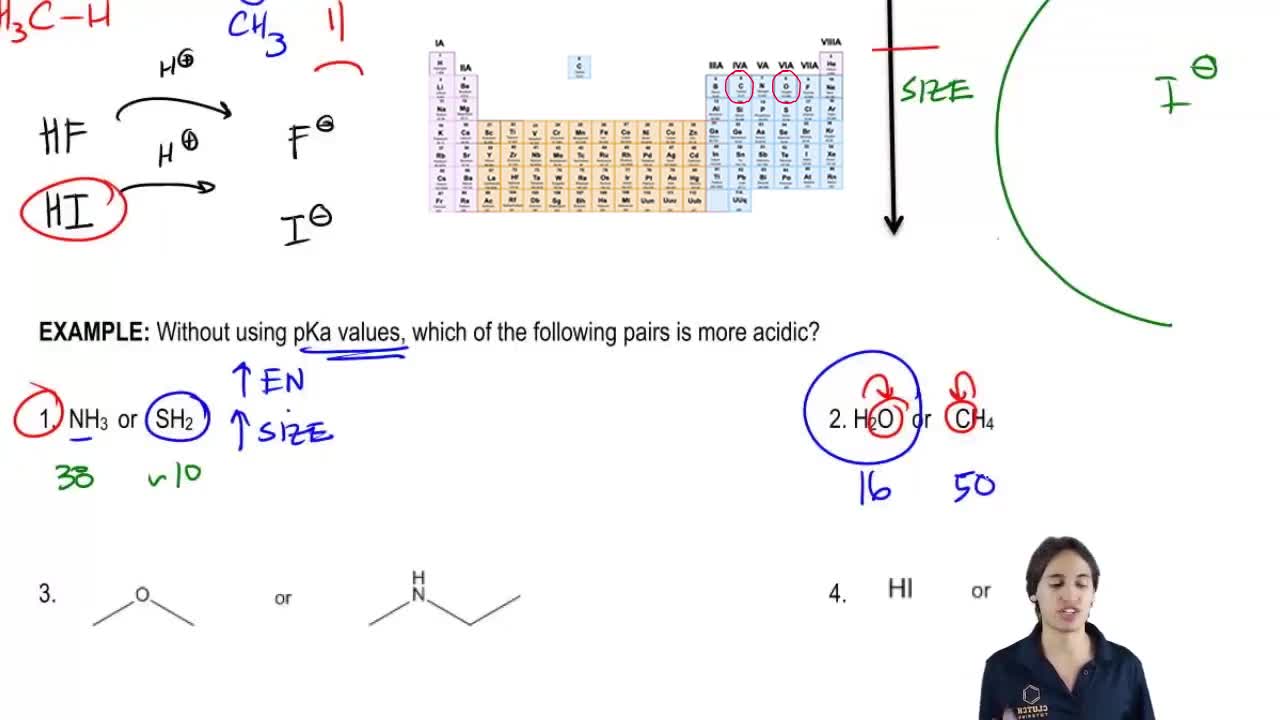
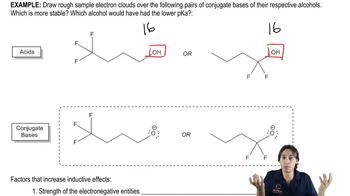
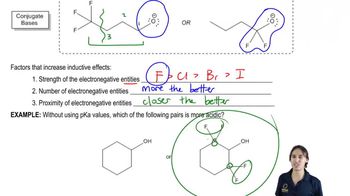
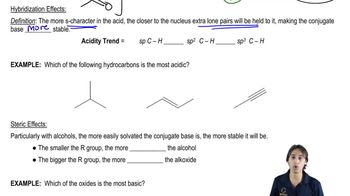
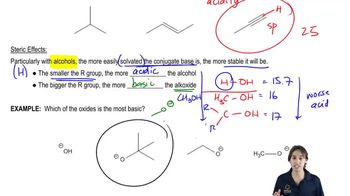
3. Acids and Bases
Ranking Acidity
Problem 11-32
Textbook Question
Textbook QuestionUse resonance forms of the conjugate bases to explain why methanesulfonic acid (CH3SO3H, pKa = - 2.6) is a much stronger acid than acetic acid (CH3COOH, pKa = 4.8).
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
99
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos