18. Aromaticity
Aromatic Hydrocarbons
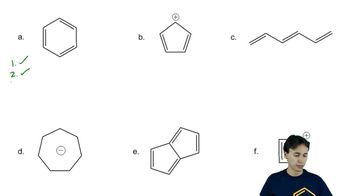
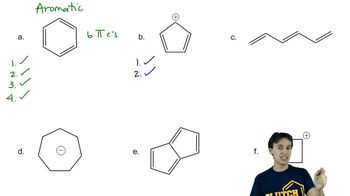
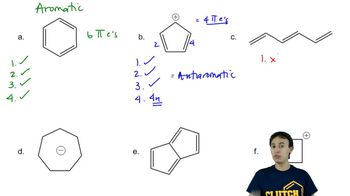
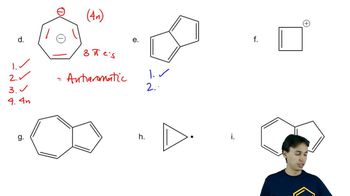
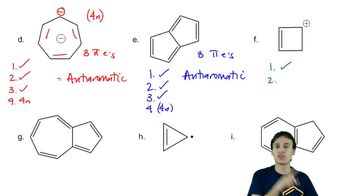
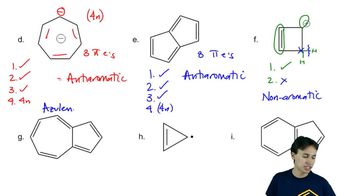
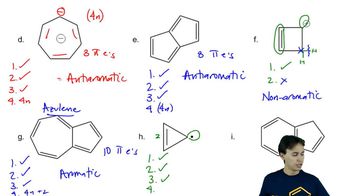
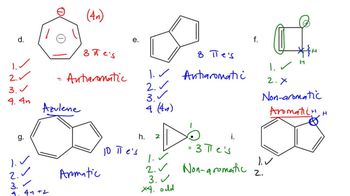
Problem 16d
Textbook Question
Textbook QuestionBiphenyl has the following structure.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
470
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos