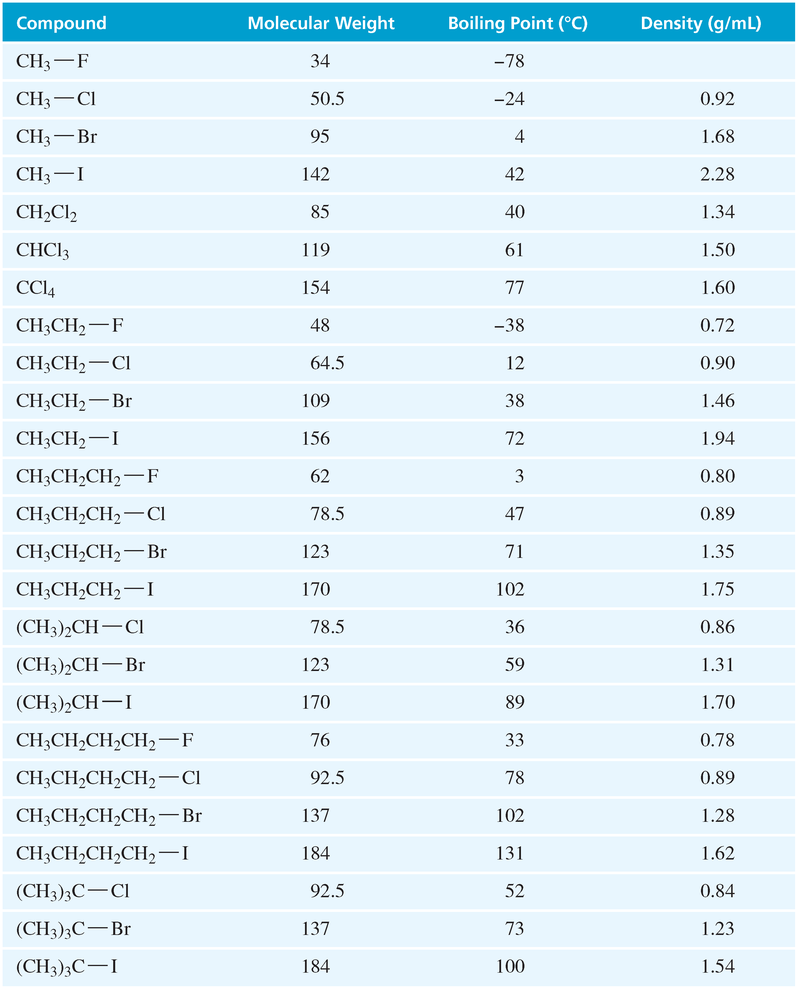
For each pair of compounds, predict which compound has the higher boiling point. Check [TABLE 6-2] to see if your prediction was right; then explain why that compound has the higher boiling point.
c. 1-bromobutane and 1-chlorobutane

 Verified step by step guidance
Verified step by step guidance

For each pair of compounds, predict which compound has the higher boiling point. Check [TABLE 6-2] to see if your prediction was right; then explain why that compound has the higher boiling point.
c. 1-bromobutane and 1-chlorobutane
Show how you might use SN2 reactions to convert 1-chlorobutane into the following compounds.
a. butan-1-ol
Show how you might use SN2 reactions to convert 1-chlorobutane into the following compounds.
b. 1-fluorobutane
The light-initiated reaction of 2,3-dimethylbut-2-ene with N-bromosuccinimide (NBS) gives two products:
a. Give a mechanism for this reaction, showing how the two products arise as a consequence of the resonance-stabilized intermediate.
For each pair of compounds, predict which compound has the higher boiling point. Check [TABLE 6-2] to see if your prediction was right; then explain why that compound has the higher boiling point.
a. isopropyl bromide and n-butyl bromide
Under appropriate conditions, (S)-1-bromo-1-fluoroethane reacts with sodium methoxide to give pure (S)-1-fluoro-1-methoxyethane.
a. Why is bromide rather than fluoride replaced?
b. Draw perspective structures (as shown on the previous page for 2-bromobutane) for the starting material, the transition state, and the product.
c. Does the product show retention or inversion of configuration? d. Is this result consistent with reaction by the SN2 mechanism?