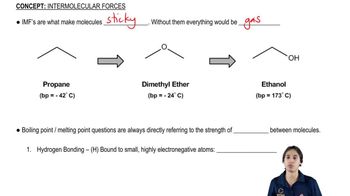
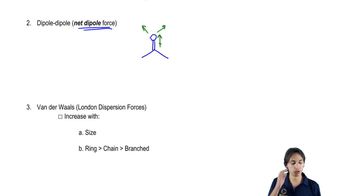
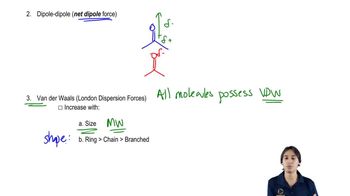
2. Molecular Representations
Intermolecular Forces
Problem 2h
Textbook Question
Textbook QuestionDiethyl ether and butan-1-ol are isomers, and they have similar solubilities in water. Their boiling points are very different, however. Explain why these two compounds have similar solubility properties but dramatically different boiling points. Diethyl ether bp 35°C 8.4 mL dissolves in 100 mL H2O Butan-1-ol bp 118°C 9.1 mL dissolves in 100 mL H2O
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
467
views
Was this helpful?
Related Videos
Related Practice