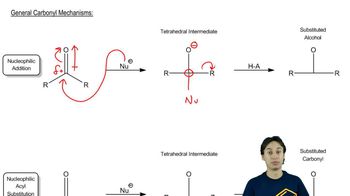
22. Carboxylic Acid Derivatives: NAS
Carboxylic Acid Derivatives
Problem 20
Textbook Question
Textbook Question(c) Peroxyacetic acid (bp = 105 °C) has a lower boiling point than acetic acid (bp = 118 °C), even though peroxyacetic acid has a higher molecular weight. Explain why peroxyacetic acid is more volatile than acetic acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
305
views
Was this helpful?
Related Videos
Related Practice