22. Carboxylic Acid Derivatives: NAS
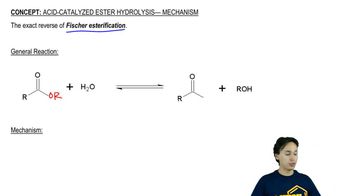
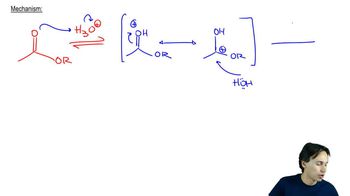
Acid-Catalyzed Ester Hydrolysis
Problem 15d
Textbook Question
Textbook QuestionUsing the mechanism for the acid-catalyzed hydrolysis of an ester as your guide, write the mechanism—showing all the curved arrows—for the acid-catalyzed reaction of acetic acid and methanol to form methyl acetate. Use HB+ and :B to represent the proton-donating and proton-removing species, respectively.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
499
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos