6. Thermodynamics and Kinetics
Entropy
Problem 4b
Textbook Question
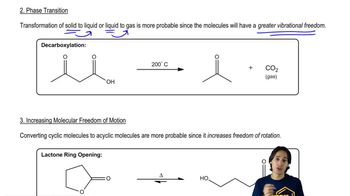
Textbook QuestionWhen ethene is mixed with hydrogen in the presence of a platinum catalyst, hydrogen adds across the double bond to form ethane. At room temperature, the reaction goes to completion. Predict the signs of ΔH° and ΔS° for this reaction. Explain these signs in terms of bonding and freedom of motion. ethene + H2 (Pt catalyst) ⇌ ethane

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
1201
views
Was this helpful?
Related Videos
Related Practice