16. Conjugated Systems
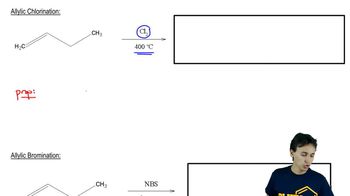
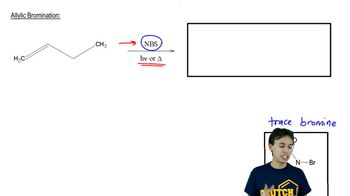
Allylic Halogenation
Problem 15-8a
Textbook Question
Textbook QuestionWhen Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% of product A and 40% of product B. When the same reaction takes place at 60 °C, the product ratio is 10% A and 90% B.
(a) Propose structures for products A and B. (Hint: In many cases, an allylic carbocation is more stable than a bromonium ion.)
(b) Propose a mechanism to account for formation of both A and B.
(c) Show why A predominates at -15 °C and B predominates at 60 °C.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
114
views
Was this helpful?
Related Videos
Related Practice