Textbook Question
Write the steps for formation of tetrachloromethane (CCl4) from the reaction of methane with Cl2 + hv.
1618
views

 Verified step by step guidance
Verified step by step guidance

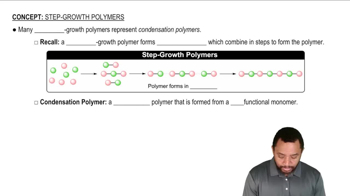

Write the steps for formation of tetrachloromethane (CCl4) from the reaction of methane with Cl2 + hv.
a. Which of the hydrogens in the structure in the margin is the easiest for a chlorine radical to remove?
b. How many secondary hydrogens does the structure have?
How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers.
a.