13. Alcohols and Carbonyl Compounds
Oxidizing Agent
Problem 11-61
Textbook Question
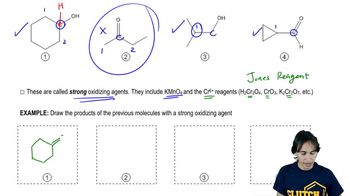
Textbook QuestionChromic acid oxidation of an alcohol (Section 11-2A) occurs in two steps: formation of the chromate ester, followed by an elimination of H+ and chromium. Which step do you expect to be rate-limiting? Careful kinetic studies have shown that Compound A undergoes chromic acid oxidation over 10 times as fast as Compound B. Explain this large difference in rates. <IMAGE of reactions>
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
132
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos