6. Thermodynamics and Kinetics
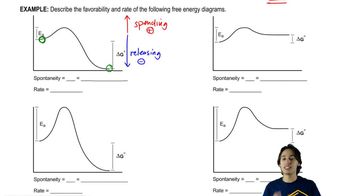
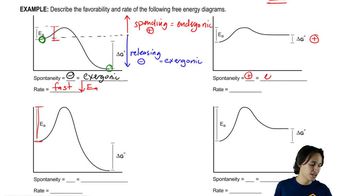
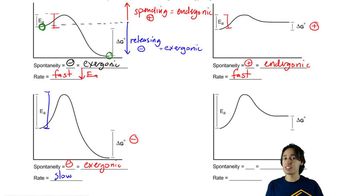
Energy Diagram
Problem 4.34a
Textbook Question
Textbook QuestionThe following reaction is a common synthesis used in the organic chemistry laboratory course.
CH3CH2CH2CH2Br 1-bromobutane + CH3O- methoxide ion —> CH3OH(methanol solvent—> CH3CH2Ch2Ch2OCH3 methoxybutane + Br- bromide ion
When we double the concentration of methoxide ion (CH3O–), we find that the reaction rate doubles. When we triple the concentration of 1-bromobutane, we find that the reaction rate triples.
a. What is the order of this reaction with respect to 1-bromobutane? What is the order with respect to methoxide ion?
Write the rate equation for this reaction.
What is the overall order?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
121
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos