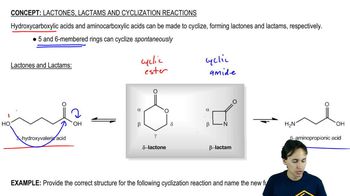
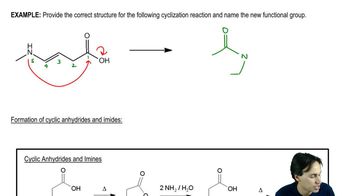
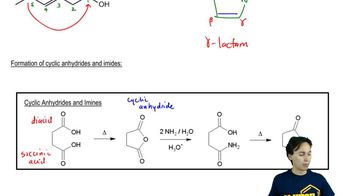
Macrolide antibiotics all have large rings (macrocycle) in which
an ester makes the ring; a cyclic ester is termed a lactone.
One example is erythromycin A, first isolated from soil bacteria
in the 1950s. Over time, some pathogenic bacteria
have developed resistance to erythromycin by evolving
an enzymatic mechanism to cleave the macrocycle
at the ketone.
To counter this resistance, chemists modified the erythromycin
structure to replace the ketone with an amine that the bacteria
could not detoxify. This modified antibiotic, azithromycin,
trade name Zithromax®, is one of the most prescribed
drugs in the world for respiratory infections.
<IMAGE>
(a) Identify the lactone group in each structure that merits the classification as macrolides.
(b) Two groups are circled. What type of functional group
are they? Explain.
 Verified Solution
Verified Solution