26. Amines
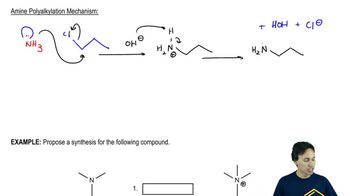
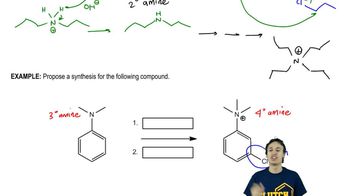
Amine Alkylation
Problem 10c
Textbook Question
Textbook QuestionDimethylamine, (CH3)2NH, has a molecular weight of 45 and a boiling point of 7.4 °C. Trimethylamine, (CH3)3N, has a higher molecular weight (59) but a lower boiling point (3.5 °C). Explain this apparent discrepancy.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
293
views
Was this helpful?
Related Videos
Related Practice