6. Thermodynamics and Kinetics
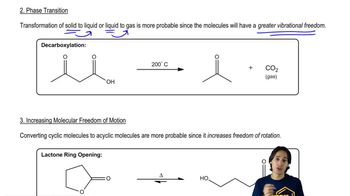
Entropy
Problem 53
Textbook Question
Textbook QuestionWhen ethene is treated in a calorimeter with H2 and a Pt catalyst, the heat of reaction is found to be -137 kJ/mol (-32.7 kcal/mol), and the reaction goes to completion.
When the reaction takes place at 1400°K, the equilibrium is found to be evenly balanced, with Keq=1.
Compute the value of ΔS for this reaction.
CH2=CH2 + H2 Pt catalyst⇌ CH3—CH3
ΔH= -137 kJ/mol (-32.7 kcal/mol)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
159
views
Was this helpful?
Related Videos
Related Practice