3. Acids and Bases
Acid Base Equilibrium
Problem 2e
Textbook Question
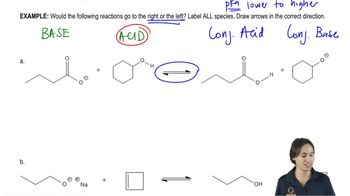
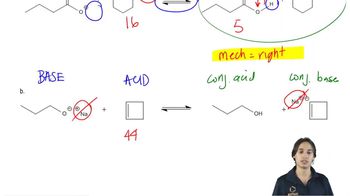
Textbook Questiona. For each of the following pairs of reactions, indicate which one has the more favorable equilibrium constant (that is, which one most favors products): 1. CH3CH2OH+NH3⇌CH3CH2O−++NH4 or CH3OH+NH3⇌CH3O−++NH4 2. CH3CH2OH+NH3⇌CH3CH2O−++NH4 or CH3CH2OH+CH3NH2⇌CH3CH2O−+CH3N+H3

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
259
views
Was this helpful?
Related Videos
Related Practice