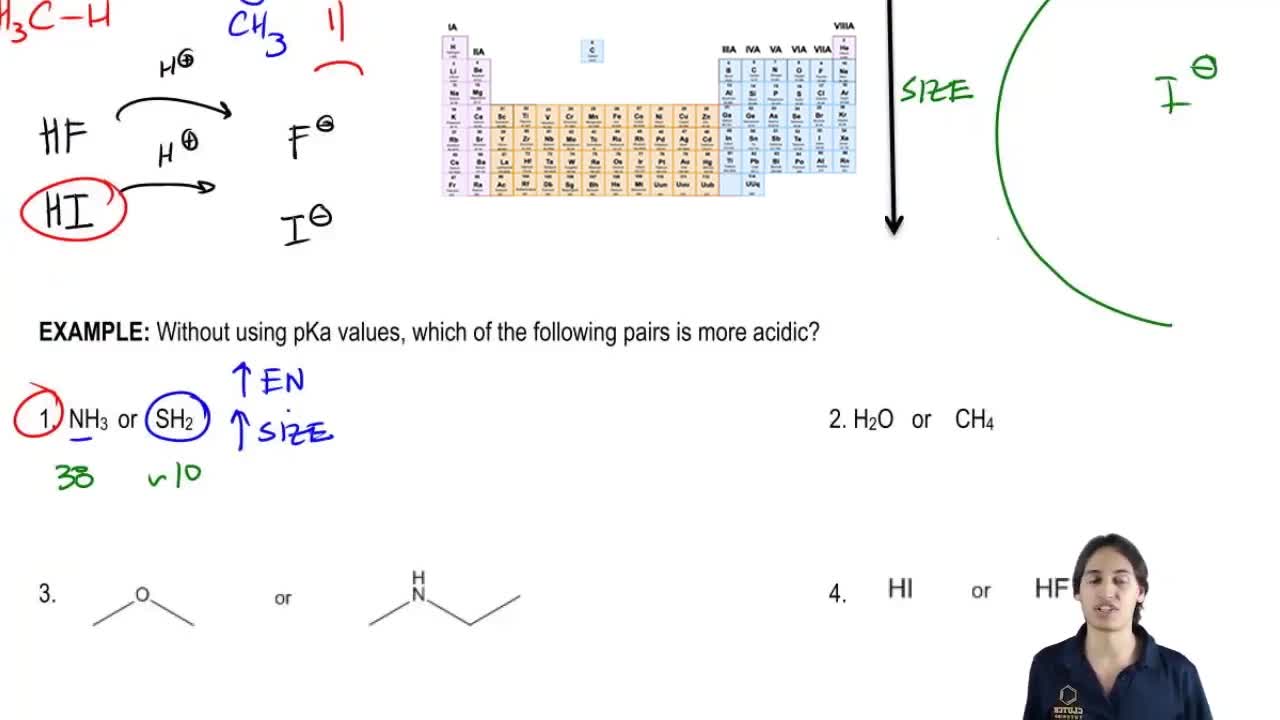
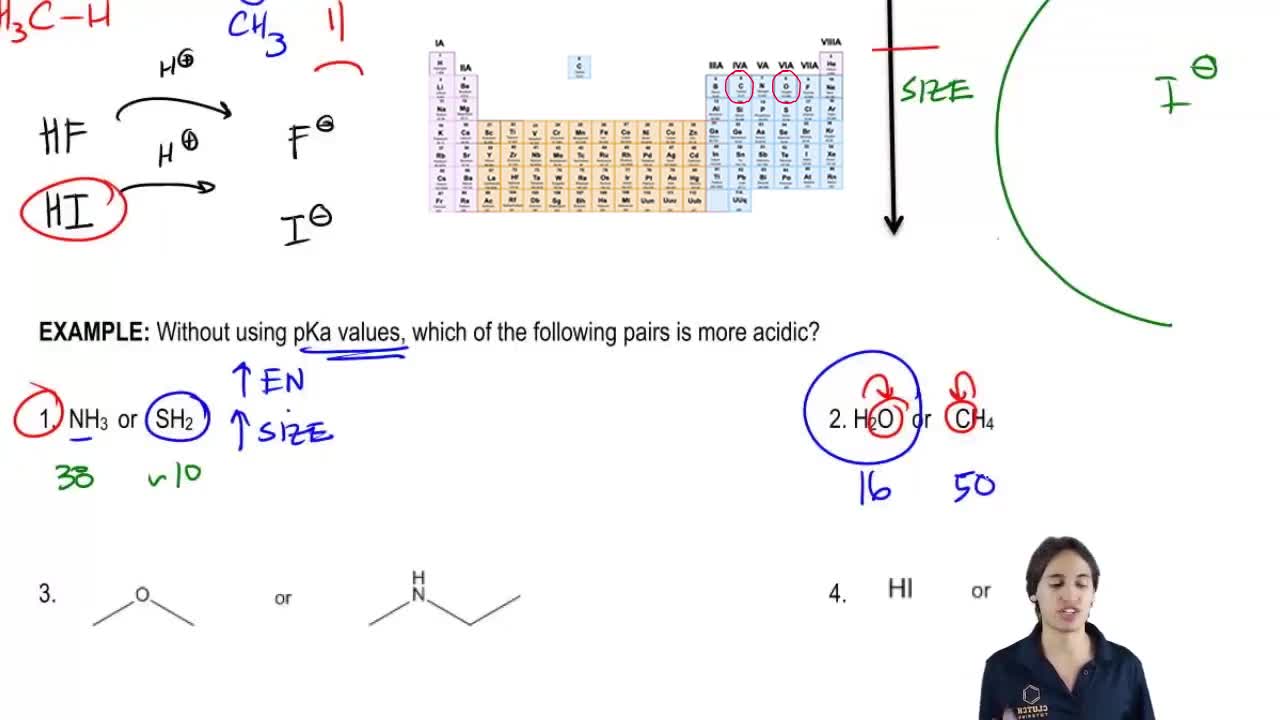
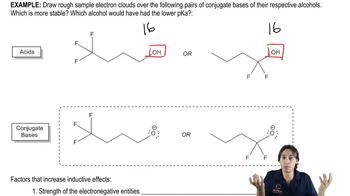
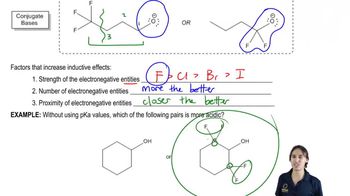
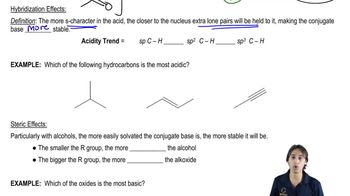
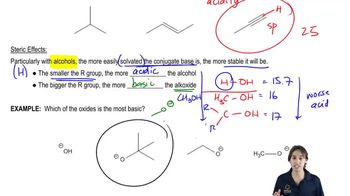
3. Acids and Bases
Ranking Acidity
Problem 10
Textbook Question
Textbook QuestionA nitro group (¬NO2) effectively stabilizes a negative charge on an adjacent carbon atom through resonance:



 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
1322
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos