3. Acids and Bases
Acid Base Equilibrium
Problem 11b
Textbook Question
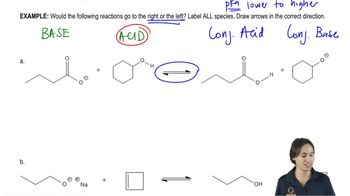
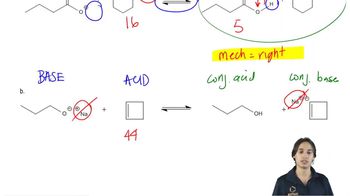
Textbook QuestionWhich of the following reactions favor formation of the products? (For the pKa values necessary to solve this problem, see Appendix I. Recall that the equilibrium favors formation of the weaker acid; see Section 2.5.) CH3MgBr + H2O <--> CH4 + HOMgBr CH3MgBr + CH3OH <--> CH4 + CH3OMgBr

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
173
views
Was this helpful?
Related Videos
Related Practice