Now before we can talk about metallic characteristics of the elements in the periodic table, it's first important to talk about the periodic trends themselves. Now the periodic trends are specific patterns in the property of elements based on their changing atomic numbers. We're going to say as we examine these periodic trends, we will examine these patterns while moving to the top right corner of the periodic table. So we're generally going to be moving this way to the top right corner of the periodic table when discussing the different types of periodic trends that exist. So keep that in mind as we investigate each and every one.
Periodic Trend: Metallic Character - Online Tutor, Practice Problems & Exam Prep
Metallic Character deals with how easily an electron can be removed from an element.
Metallic Character
Periodic Trend: Metallic Character
Video transcript
Periodic Trend: Metallic Character
Video transcript
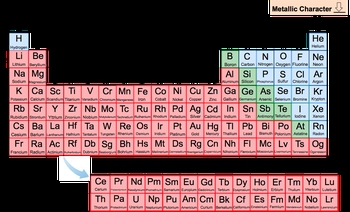
Now metallic character has to do with how easily an element can lose an electron. The easier it is for an element to lose an electron, the greater its metallic character. Now remember, metals tend to lose electrons, nonmetals tend to gain electrons. When it comes to the major periodic table classifications, we have our metals, our metalloids, and our nonmetals. And we're going to say the periodic trend here is that metallic character is going to decrease as we move from left to right across a period, so as we're going this way, and as we go up a group. This makes sense because where are the metals on the periodic table? They're all clustered more to the left and the bottom. As we're heading towards the top right corner, we're moving away from the metals, so our metallic character is supposed to decrease. The closer you are to the metals, the more metallic character you have. The further away you are from the metals, the less metallic character you're going to have.
Moving towards the top right corner of the Periodic Table causes metallic character to decrease.
Periodic Trend: Metallic Character Example 1
Video transcript
So here it says, based on the periodic trend, which element would have the greater metallic character? So here, let's compare sulfur versus tellurium. Looking on the periodic table, we see sulfur here, we see tellurium here. Remember, the trend is as we head towards the top right corner, our metallic character will decrease, and as we move up a group, our metallic character will also decrease. So since Tellurium is further down, it should have more metallic character, it's closer to the metals. As we're going up the group, we're becoming less metallic and therefore sulfur would have a lower metallic character. So just remember, the closer we get to the metals, the greater our metallic character will be. The further we get from the metals, the lower our metallic character will be.
Between which two elements is the difference in metallic character the greatest?
