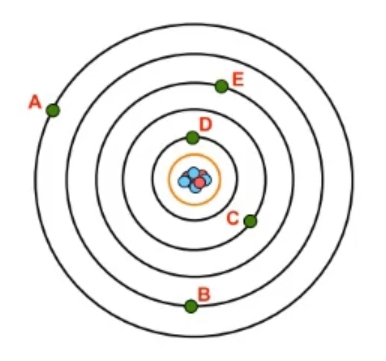
Now recall a shell, which uses the variable \( n \), is a grouping of electrons surrounding the nucleus that ties into their potential energy, so their energy of position. Now we're gonna say as the value of \( n \) increases, then both the size and energy level of an atomic orbital will increase. And we're going to say here that the energy levels, also the shell numbers of an atom can be tied to the periods or rows of the periodic table. So here we have an atom. This atom has its nucleus, and we can see that I have given it 7 shells. Right? Remember, each black orbital represents a shell. 1st shell all the way to the 7th shell here. We'll realize here that the shells of an atom are directly related to the periods or row of the periodic table. So remember, your periods or rows of periodic table go from left to right. So this is row 1, so this is shell 1, shell 2, 3, 4, 5, 6, and 7. Now currently there are 7 rows in the periodic table, but what you need to understand about the periodic table is that it is dynamic. A lot of these elements found in the 7th row of the periodic table have been discovered within the past decade. A bunch of these here were formally named in the past 10 years. So theoretically, as we move to new planets, as we explore more of the earth, as our technology gets more advanced, we're gonna create and find new elements. So eventually, there's gonna be an 8th row of elements, and then a 9th row, and so on and so forth. We're only limited by our imagination and our ingenuity. The number of elements can continuously increase again as we discover them and as we create them. Now we're gonna say because of this, the limitation of the \( n \) value is that it must be an integer, remember, whole number, from 1, because the smallest number shell can be is 1, to infinity. There's an infinite number of possible shells. Again, we're only limited by our resources, our imagination, and our ingenuity. We can create new elements with 10 shells, 12 shells, and so on. So keep in mind the relationship between rows of the periodic table and shell numbers of an atom and realize the limitation of it is that it can be any number from 1 to infinity.
Electronic Structure: Shells - Online Tutor, Practice Problems & Exam Prep
Shell number gives both the size and energy of the shell.
Electronic Structure:Shells
Electronic Structure: Shells Concept 1
Video transcript
Electronic Structure: Shells Example 1
Video transcript
Of the following, is a possible value for the shell of an atom? Remember, we talked about the only real limitation when it comes to the shell number of an atom is that it's connected to the rows of the periodic table, and because of that it can be any integer from 1 to infinity. So, negative 3 can't be an exact number because that's less than 1. Same thing with negative 4. It has to be a number from 1 to infinity, so 0 is out, and e is out. So n being 2, meaning the second shell of an atom, is a possible value for n. Right? So here, the only option that works is option d.
Which of the following shell number values is a possible value for the element highlighted?
Which electron possesses the lowest possible energy from the image provided?