Now recall that stoichiometry deals with the numerical relationship between compounds in a balanced chemical equation. Now with a thermochemical equation, we're going to deal with chemical reactions that include an enthalpy of reaction, which is ΔHrxn, so delta H of reaction. Here with thermochemical equations, we're going to be introduced to our thermochemical stoichiometric chart. Now here, the chart uses the given quantity of a compound to determine the unknown quantity of another compound. So here with the thermochemical equation, we have our balanced chemical equation, and to the side of it, you'll see your delta H of reaction. It's our job to make a connection between your enthalpy of reaction and either moles, grams, molecules, what have you, in terms of the chemical reaction. So in a thermochemical equation, what we're trying to do is not a mole-to-mole comparison; we're trying to do a delta H to mole comparison. And that's the key difference with our thermochemical equation. Now that we've seen this, let's move onwards and talk about more, with thermochemical equations.
Thermochemical Equations - Online Tutor, Practice Problems & Exam Prep
Thermochemical Equations involve a balanced chemical equation with a given enthalpy value.
Thermochemical Equations
Thermochemical Equations
Video transcript
Thermochemical Equations
Video transcript
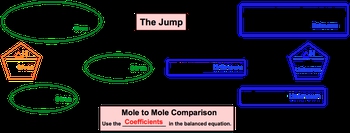
Now, if you've seen my stoichiometric chart under stoichiometry, or solution chemistry, or gas stoichiometry, this should be pretty familiar to you. But if this is the first time you're seeing this stoichiometric chart, let's go through it. Alright. So what we need to realize here is that with thermochemical equations, it's pretty common to be given the ΔH of our chemical reaction. So we'll start out with ΔH of given. And like I said before, in stoichiometry we're used to doing a multiple comparison which can still happen here, but the more important thing is that we establish a connection between the ΔH of reaction and one of the moles for one of the compounds within our chemical, reaction. So, here we go from ΔH of given to moles of given. And what we can say here is that besides going from ΔH of given to moles of given, we can go from grams of given to moles of given, or we can go from ions, atoms, formula units, or molecules of given to moles of given. Once we get there, we have to go to moles of our unknown. To do this requires a leap of faith in a sense, because you're going from an area where you know information to an area where you know nothing at all. So, we call this the jump. When you make this jump, in order to do it correctly you have to do a mole to mole comparison and use the coefficients in the balanced equation. From this point, you are at moles of unknown, and from here you can go in any way you want. You can go from moles of unknown to ions, atoms, formula units, or molecules, you can go to grams, or you can go to a new ΔH of unknown. So, just realize with the thermochemical equation, we'll have a balanced chemical equation, which introduces the idea of stoichiometry, but for it to be a thermochemical equation, we'll also have the ΔH of reaction present.
A modified version of the stoichiometric chart can used for thermochemical equations.
Thermochemical Equations Example 1
Video transcript
Here it says, consider the following thermochemical reaction. We have 2 moles of magnesium solid reacting with 1 mole of oxygen gas to produce 2 moles of magnesium oxide solid. It gives us an enthalpy of reaction equal to negative 1204 kilojoules. Here we're asked how many grams of magnesium oxide are produced during enthalpy change of negative 375 kilojoules. So what we have to do here is we have to convert the given quantity into moles of given. Alright. So they're giving us negative 375 kilojoules of energy, and what we need to do is establish a relationship between magnesium oxide and this value of negative 375. Well according to my balanced equation, for step 2, it says we need to do a mole to mole comparison to convert moles of given into moles of unknown. Here because it's a thermochemical equation, it's going to be moles of given relating to delta h of reaction. So we're going to say for every 2 moles of magnesium oxide, the energy involved or enthalpy involved is negative 1204 kilojoules per mole. So we've just found our moles in terms of magnesium oxide.
Steps 3 says, if necessary convert the moles into desired units. Here they want grams not moles, so we're going to do one more step and say for every one mole of magnesium oxide, the mass of magnesium oxide, 1 magnesium, is 24.31 grams according to the periodic table. One oxygen is 16 grams. Multiplying and adding those numbers together gives us 40.31 grams. Moles cancel out and now I'm going to have 25.11 grams of magnesium oxide. Step 4 isn't needed here because, in step 4, if we have to calculate more than one final amount then we must compare them to determine the theoretical yield. Here we're only given one given the amount of negative 375 kilojoules and using that helped us to determine the final answer of 25.11 magnesium oxide, grams of magnesium oxide.
Nitromethane (CH3NO2), sometimes used as a fuel for drag racing, burns according to the following reaction:
4 CH3NO2 (l) + 7 O2 (g) → 4 CO2 (g) + 6 H2O (g) + 4 NO2 (g) ∆Hº = – 2441.6 kJ
How much heat is released by burning 125.0 g of nitromethane (MW:61.044 g/mol)?
Consider the following reaction:
2 C6H6 (l) + 15 O2 (g) → 12 CO2 (g) + 6 H2O (g) ∆Hº = – 6278 kJ
What volume of benzene (C6H6, d = 0.880 g/mL, molar mass = 78.11 g/mol) is necessary to evolve 5.19 x 109 kJ of heat?
1.47 x 108 mL
5.19 x 109 mL
4.37 x 107 mL
9.51 x 109 mL
The creation of liquid methanol is accomplished by the hydrogenation of carbon monoxide:
CO (g) + 2 H2 (g) → CH3OH (l) ∆Hº = – 128.1 kJ
How much heat (in kJ) is released when 125.0 g CO reacts with 2.32 x 102 g H2?
Your Introduction to Chemistry tutor
- The following equation shows the conversion of aluminum oxide (from the ore bauxite) to aluminum: 2 Al2O3(s) ...
- The vaporization of Br2 from the liquid to the gas state requires 7.4 kcal/mol (31.0 kJ/mol). How many kilojo...
- Converting liquid water to solid ice releases 1.44 kcal/mol (6.02 kJ/mol). How many kilocalories are released...
- Glucose, also known as 'blood sugar' when measured in blood, has the formula C6H12O6. What is the minimum amo...
- For the production of ammonia from its elements, ∆H = -22 kcal/mol(-19 kJ/mol). How much energy (in kilocalor...
- For the evaporation of water, H2O(l) → H2O(g), AT 100°C, ∆H = +9.72 kcal/mol (+40.7 kJ/mol). How many kilojo...
- Methanol, CH3OH, is used as race car fuel. How many kilojoules are released by burning 50.0 g of methanol?
