Hey guys, in this new video, we're going to take a look at positron emission. So, here we're going to say positron emission occurs when an unstable nucleus emits a positron. Now, what's a positron? A positron is an antiparticle of the electron. Remember, the electron is represented by e-. A positron is the opposite of that. So, it looks like an electron, but instead of it having a negative sign, it will have the opposite sign. So, it will be a positive electron. So, a positron is considered just a positive electron. I know this is weird, but again remember, we're dealing with nuclear reactions. So, a lot of unaccustomed things that we are not used to seeing do occur. And one of them is this positron. So, we're going to say here, here's our positron. Now, because we're talking about the word emission again, emission would mean decay, which means that this positron would be a product. So, let's think of an example. Here, Einstein has his own element named after him, Einsteinium. So Einsteinium will deal with isotope 253 of Einsteinium. So, Einsteinium is ES on our periodic table. It has an atomic number of 99. We're going to emit a positron and because we're emitting a positron, let's see. So because the atomic mass is 0, the new element is still going to be 253. But because the bottom is 1, what number plus 1 gives me 99? It would have to be 98. So, here, that would just be CF. So, this would be an example of a positron decay or positron emission.
Positron Emission - Online Tutor, Practice Problems & Exam Prep
A positron emission or positron decay occurs when an unstable nucleus ejects a positron particle to create a new element.
Understanding Positron Emission
A positron particle is referred to as the anti-electron particle because it looks like a positively charged electron.

Positron Emission Concept 1
Video transcript
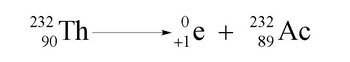
Positron Emission Example 1
Video transcript
Now, based on that, let's answer these two questions. It says, write balanced nuclear equations for each of the following positron emissions. So again, your positron will be a product. So here we're dealing with uranium 235. Uranium is U. On your periodic table, it's going to have an atomic number of 92. We emit a positron, so this is going to be:
U 235 , 92 → Pa 235 , 91 + e + 0 , 1The next one is radon which is Rn. Radon has an atomic number of 86. We emit a positron. So now, the element is still going to be 222 and 85 +1 gives me the 86. So, that's Actinium. The equation is:
Rn 222 , 86 → Ac 222 , 85 + e + 0 , 1These would be two examples of our positron emission.
Positron Emission Example 2
Video transcript
Now, following what we've learned so far about all the different types of decays and emissions out there, let's try our best to answer this one. So here we're going to say a nucleus of Thorium-225 undergoes three alpha decays, four beta decays, and a gamma emission. What is the product? So, remember Thorium has an atomic number of 90. It's going to undergo three alpha decays, and alpha decays basically produce the helium or alpha particle, plus four beta decays, so that's four electrons being emitted, plus gamma, which is 0 over 0. So, basically when you have three helium particles being emitted, that's 3×4 which is 12, 3×2 which is 6, 4×0 which is 0, 4×-1 which is -4. Again, this is not the proper way to write it. I'm just combining all the math just to make it easier for us to see what the answer is. So, we're going to say 12 here. So the new element would have to be 213 because 12 + 213 will give me the 225 I started out with. And then here, 6 plus this negative 4 gives me 2. So it would have to be 88 here because 88 + 2 will give me back the 90 I started out with. So, then we're going to say we have 88, so that is Ra, which is radium. And because we underwent a gamma emission, technically, this would be in an excited state. So, we put a little asterisk there to show that it's in an excited state. So, that's all we have to do. And this top equation is the correct way to write the equation. Remember, oops, remember I just combined everything here to make the math easier for us to see what's going on. But it's this top equation that's the correct format that your professor would be looking for. So, just remember, fundamentally, decay and emission mean the particle will be a product. Capture or absorption means it'll be a reactant. That's the key to this. And then, just remembering what's an alpha particle, a beta particle, a positron, a gamma, all those different concepts.
