Hey guys, in this new video, we're going to take a look at a particular type of acids and bases. Now, we know that there are 2 types of acids. There are binary acids and oxy acids. And we know how to identify bases. Now, just realize that there are going to be 3 types of categories for acids and bases. The first one being Arrhenius. Now we're going to say the most general definition for acids and bases was developed by Arrhenius near the end of the 19th century. We're going to say according to him, we're going to say that the H+ cation and the OH- anion were the fundamental pieces that helped to identify acids and bases. But the thing with him is, his definition failed to describe what happens to acids and bases when we take them out of water. When we take them out of aqueous solutions, what happens? His definition failed to talk about that. Now, we're going to say according to his Arrhenius definition, it states that an acid is a compound that increases my concentration of H+ when dissolved in a solvent. The solvent being water. Because to him acids and bases only operated in aqueous environments. Water. So we're basically going to say an example of an Arrhenius acid is HCl. When it breaks down in water, it's going to give me H+ plus Cl-. Now, because Arrhenius's definition for an acid applies to both strong and weak acids. Because if they can produce H+ then they're technically Arrhenius. Now, so the limitation here is that to him, if you didn't have an H involved, you couldn't be an acid. But we'll learn later that this is just the wrong way of looking at things. Okay? So, now that we know the definition for an acid, let's look at the definition for a base. We're going to say according to his definition, Arrhenius definition, a base is a compound that increases the concentration of OH- when dissolved in a solvent. Now what we're going to say here is a good example we have is NaOH. When it dissolves in water, it gives me Na+ plus OH-. If it didn't create any OH- as a product then it couldn't be described as an Arrhenius base. Again, this definition is the broadest of the 3 definitions we're going to see. It was the earliest one developed that's why it's so broad. It's basically any compound with H positive or OH-. We'll learn more and more definitions that are more precise to exactly what acids and bases are. Now that we've seen this, I want you guys to attempt to do these questions. You have to tell me, how do each of these compounds break up to give me my products? And based on the products, are they Arrhenius Bases, Arrhenius Acids, or maybe neither one? Good luck on the first one guys. And again, once you're done with it, come back. Click on the explanation button and watch a video of me explaining how to break it up and what exactly it is that we're breaking up.
14. Acids and Bases
Arrhenius Acid and Base
14. Acids and Bases
Arrhenius Acid and Base - Online Tutor, Practice Problems & Exam Prep
The most general definition for acids and bases was developed by Svante Arrhenius near the end of the 19th century.
Understanding Arrhenius Acids & Bases
According to Svante Arrhenius the two most important ions fundamental to the concept of acids and bases were H3O+ , the hydronium ion, and OH –, the hydroxide ion.
1
concept
Arrhenius Acids & Bases Concept 1
Video duration:
3mPlay a video:
Video transcript
An Arrhenius acid increases the H+ ion when dissolved in a solvent.
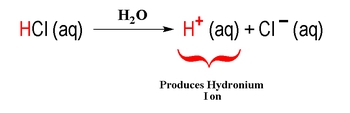
An Arrhenius base increases the OH – ion when dissolved in a solvent.

2
Problem
ProblemWhich ions are formed from the dissociation of the following compound?
A
Sr(OH)2 (s) → Sr2+ (aq) + 2 OH-
B
Sr(OH)2 (s) → Sr2+ (aq) + OH-
C
Sr(OH)2 (s) → 2 Sr+ (aq) + OH-
D
Sr(OH)2 (s) → 2 Sr+ (aq) + 2 OH-
3
Problem
ProblemWhich ions are formed from the dissociation of the following compound?
A
B
C
D
No reaction
4
Problem
ProblemWhich ions are formed from the dissociation of the following compound?
A
B
C
D
Your Introduction to Chemistry tutor
Additional resources for Arrhenius Acid and Base
PRACTICE PROBLEMS AND ACTIVITIES (3)







