9. Electrons in Atoms and the Periodic Table
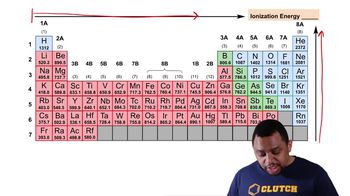
Periodic Trend: Ionization Energy (Simplified)
Problem 73
Textbook Question
Textbook QuestionWhich statements completed with a to e will be true and which will be false? An atom of N compared to an atom of Li has a larger (greater) b. ionization energy
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
271
views
Was this helpful?
Related Videos
Related Practice