8. Quantities in Chemical Reactions
Stoichiometry
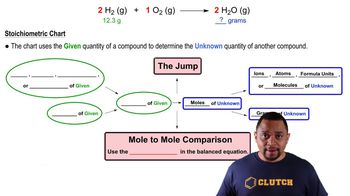
Problem 58
Textbook Question
Textbook QuestionAlthough Cu is not sufficiently active to react with acids, it can be dissolved by concentrated nitric acid, which functions as an oxidizing agent according to the following equation: Cu(s) + 4 HNO3(aq) → Cu(NO3)2(aq) + 2 NO2(g) + 2 H2O(l) Is 35.0 g of HNO3 sufficient to dissolve 5.00 g of copper?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
351
views
Was this helpful?
Related Videos
Related Practice