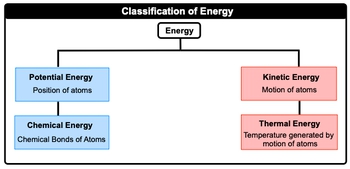
Thermochemistry is the study of matter and energy associated with chemical reactions or physical changes. Energy itself is just the capacity to do work or to produce heat. Now when we talk about energy, realize that there are different types of energy, but in this chapter, we're only going to focus on a selected few. Now when we take a look at the big picture, energy itself, we can say here that it can be broken down initially into either the position of atoms or the motion of atoms. When we're talking about the position of atoms, this is just simply our potential energy, our energy of position. And then if we're talking about the movement or motion of atoms, this is connected to kinetic energy. Now both potential energy and kinetic energy can be further broken down into other types of energy. Potential energy, we can connect it to another one which deals with the chemical bonds of atoms. This is just simply chemical energy. And then kinetic energy can be broken down further into energy associated with temperature generated by motion of atoms. So this would be called thermal energy. So just remember, energy is just the capacity to work and to produce heat. And when we're talking about energy in this chapter, we're mainly concerned with these different types of energy forms. Now, of course, there are other types of energy that exist. We'll go into those in later chapters, but for now just remember these particular four.
Nature of Energy - Online Tutor, Practice Problems & Exam Prep
Thermochemistry is the study of matter and energy associated with chemical reactions or physical changes.
Classification of Energy
Nature of Energy
Video transcript
Nature of Energy
Video transcript
In our discussion of the concept of energy, it's important to remember that the SI unit for energy is Joule, and it's named after the English scientist James Joule. When it comes to joules, we're going to say that there are 3 particular types of conversion factors associated with it.
We're going to say here for the first one, we have one calorie which is lowercase c, this equals 4.184 joules. Next, we have 1 capital C calorie, this one is associated with food nutrition. This particular calorie equals 4184 joules. And then finally, kilowatt-hours. Usually, when we talk about an electrical bill, it's associated with kilowatt-hours. So we're going to say here kilowatt-hours equals 3.6×106 joules.
These are 3 conversion factors associated with our SI unit for energy. Also realize here that I didn't put purple boxes around them, so usually, you're not expected to memorize them. They're often given to you within the question or they're given to you on a formula sheet when taking an exam. But here, just again remember, these are 3 common types of conversion factors associated with joules.

Nature of Energy Example 1
Video transcript
Here it states, which of the following statements deals with potential energy without non-chemical energy associated with it. Remember, potential energy is just the energy of position, whereas chemical energy, which is an offshoot of potential energy, is just associated with the chemical bonds of atoms. Alright. So if we take a look here, it says a car traveling with a velocity of 51 meters per second with a mass of 1,250 kilograms. Here we're talking about velocity, we're talking about the motion of this car, so this is more associated with kinetic energy. So, this is out.
Next, your chemistry book sitting on a table counter near the trash can, weighing 12 newtons at a height of 1.2 meters. So make sure it doesn't fall into the trash can. Here, they're just talking about the position of the chemistry book. It's at a certain height, and we're talking about a force on it. Here, this is purely potential energy, so this would be our answer. But let's look at the other options.
A chunk of coal being thrown into a furnace to generate heat. Here we're talking about heat being generated from this. This is closely related to thermal energy. And then next, we have the warmth coming from a campfire. So again, we're talking about temperature, we're talking about heat. So this is a version of kinetic energy in the form of thermal energy.
So, out of all the choices, the only one that is purely potential energy without talking about chemical energy would have to be option B.
An energy efficient refrigerator uses 780 kWh of electrical energy per year. How many kilocalories of electricity does it use in three years?
2.0 x 105 kcal
8.3 x 105 kcal
2.0 x 106 kcal
8.3 x 106 kcal
Do you want more practice?
Your GOB Chemistry tutor
- Discuss the changes in the potential and kinetic energy of a roller-coaster ride as the roller-coaster car cli...
- Indicate whether each of the following statements describes potential or kinetic energy: c. the energy in a l...
- Indicate whether each of the following statements describes potential or kinetic energy: b. kicking a ball
- Using the energy values for foods (see TABLE 3.7), determine each of the following (round off the answer for e...
- Using the energy values for foods (see TABLE 3.7), determine each of the following (round off the answer for e...
- For dinner, Charles had one cup of clam chowder, which contains 16 g of carbohydrate, 12 g of fat, and 9 g of ...
- A patient receives 3.2 L of intravenous (IV) glucose solution. If 100. mL of the solution contains 5.0 g of gl...
- What is a catalyst, and what effect does it have on the activation energy of a reaction?
- If a catalyst changes the activation energy of a forward reaction from 28.0 kcal/mol to 23.0 kcal/mol, what ef...
- Using energy values from TABLE 3.8, determine each of the following: d. If expending 3500 kcal is equal to a ...
- Using energy values from TABLE 3.8, determine each of the following: c. If Charles consumes 1800 kcal per day...
- b. For the amount of exercise that Charles did for one week in part a, if expending 3500 kcal is equal to a lo...
- For the unbalanced combustion reaction shown, 1 mol of ethanol, C2H5OH, releases 327 kcal (1370 kJ): C2H5OH ...
- A 70.0-kg person had a quarter-pound cheeseburger, french fries, and a chocolate shake. (3.5) d. Using TABL...
- The relationship between the nutritional unit for energy and the metric unit is 1 calorie = 1 kcal. a. One don...
- When a 0.66-g sample of olive oil is burned in a calorimeter, the heat released increases the temperature of 3...
- When 1.0 g of gasoline burns, it releases 11 kcal. The density of gasoline is 0.74 g/mL. (3.4, 3.6) b. If a t...
- A patient is receiving 3000 mL/day of a solution that contains 5 g of dextrose (glucose) per 100 mL of solutio...
