Gibbs free energy, which is Δg, is a measure of energy change of a chemical or physical process that can be used to do work. Now we're gonna say the sign of Δg determines the spontaneity of a reaction. Alright. So we're gonna say when our Δg value is less than 0, meaning that it's negative, that means our reaction is spontaneous. When it's the opposite, when Δg is greater than 0, so it's positive, it becomes non spontaneous. And then finally, when Δg is equal to 0, that means we are at equilibrium. So we're neither spontaneous nor non spontaneous. We're right on the border. So just remember the sign of Δg can determine the spontaneity of a chemical reaction.
Gibbs Free Energy (Simplified) - Online Tutor, Practice Problems & Exam Prep
Gibbs Free Energy represents energy associated with a chemical reaction that can be used to do work.
Gibbs Free Energy
Gibbs Free Energy (Simplified) Concept 1
Video transcript
Gibbs Free Energy (Simplified) Example 1
Video transcript
If ΔG is small and positive, which of the following statements is true? Before we solve this question, realize that we're dealing with reversible reactions, so they can go in the forward direction or the reverse direction. Now, they're telling us ΔG is small, so that means it's close to 0, and it's positive. That means it's slightly greater than 0, which means it's non-spontaneous. When we say that, we mean non-spontaneous in the forward direction. But remember, we're dealing with reversible reactions when we talk about Gibbs free energy. So, if you're non-spontaneous in the forward direction, that means you are spontaneous in the reverse. So, let's see what makes the most sense.
It's positive, so that means it's going to be non-spontaneous in the forward, which means that options A and B are out because they wouldn't be spontaneous in the forward. However, it will be spontaneous in the reverse, as stated. But now, is it far from equilibrium or near equilibrium? Remember, when ΔG is equal to 0, we are at equilibrium. Since ΔG is small, that means it's a number that's pretty close to 0. This implies that we would be near equilibrium. This would mean that option D is the correct answer.
Gibbs Free Energy (Simplified) Concept 2
Video transcript
When the sign of Δg is unknown, we can say that the spontaneity of a reaction can be predicted from the signs of enthalpy, which is Δh, and entropy, which is Δs. So here we're going to say that when they're both positive, we are spontaneous at high temperatures. And if both of them being positive makes it spontaneous at high temperatures, then what happens when it's the opposite? Well, when they're both negative, we'd be spontaneous at low temperatures. Next, we're going to say when Δh is positive and Δs is negative, you are always non-spontaneous in terms of your chemical reaction. And if we do the opposite of that, if Δh is now negative and Δs is positive, then you'd be spontaneous for your chemical reaction. So, just remember, the easiest way to remember this, here your Δh, positive and negative. Here your Δs, positive and negative. So you'd say high t, low t, non, and spawn. So the signs of our Δh and Δs for a chemical reaction can be used to determine the right temperature conditions to make it spontaneous or not.
Gibbs Free Energy (Simplified) Example 2
Video transcript
Here we're going to say that PCl3, phosphorus trichloride, plus chlorine gas react together to give us phosphorus pentachloride. At 25 degrees Celsius, the enthalpy is negative 92.50 kilojoules. Which of the following statements are true? This is an endothermic reaction. Well, ΔH here is negative, so it is exothermic, not endothermic. If the temperatures increase, the ratio of our products over reactants will increase.
First of all, we're going to say products over reactants is talking about our equilibrium expression. So, we're talking about our equilibrium constant K. Next thing we're going to say is, let's determine the sign of ΔH and ΔS. We know that ΔH is negative because we're told that right from the beginning, so we're dealing with this one. And then what you're going to say, you can determine the sign of ΔS. We have 2 reactants combined to give us one product, so we're forming bonds, which means that ΔS is negative. So that means we fit in this slot, which means that as temperatures decrease we become more spontaneous. Okay. So we're spontaneous at low temperatures.
So if we're increasing the temperature, that's going to have the opposite effect. It's going to make us less spontaneous and more non-spontaneous. And realize when we're making a reaction more non-spontaneous, that means it doesn't want to happen. That means the reverse direction is favored. So that means that reactants are favored more than products. K. So remember K equals products over reactants. We're heading in the reverse direction where reactants become more favored, so your bottom part is becoming bigger and as a consequence, your top part is becoming smaller. So overall, what's happening to K? K would be decreasing, so this is not true. Here the ΔS for the reaction is negative? Yes. When multiple reactants combined give me one product? That is a decrease in entropy, so it's negative. And ΔG for the reaction has to be negative at all temperatures? No. It's only going to be negative or spontaneous at low temperatures. So the only statement here that's true would be option c.
What are the signs of ∆H, ∆S and ∆G for the spontaneous conversion of a solid into gas?
You calculate the value of ΔG for a chemical reaction and get a positive value. Which would be the most accurate way to interpret this result?
If a mixture of reactants and products is created and left to equilibrate, the equilibrium mixture will contain more reactant than product.
If a mixture of reactants and products is created, we cannot say anything about its composition at equilibrium but we can say it will reach equilibrium very rapidly.
The reaction will not occur under any circumstances.
If a mixture of reactants and products is created and left to equilibrate, the equilibrium mixture will contain more product than reactant.
Consider the combustion of butane gas and predict the signs of ΔS, ΔH and ∆G.
C4H10(g) + 13/2 O2(g) ⟶ 4 CO2(g) + 5 H2O(g)
Gibbs Free Energy (Simplified) Concept 3
Video transcript
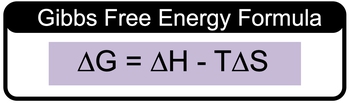
We can calculate the value for Gibbs free energy, delta g. We're going to say we do this by using Gibbs free energy formula. It allows us to calculate the value of delta g by using delta h, usually, it's given to us in kilojoules, delta s, which is usually given to us in joules per Kelvin, and temperature, which is given to us in Kelvin. Together, it gives us the formula Δg=Δh−tΔs. Now remember, it's always important to include your units within your calculations. And realize that if they give us delta h in kilojoules and delta s in joules, those units do not match. So, you have to convert them so that they do match. So either make both units kilojoules or you make both units joules. What's customarily done is that we convert everything to kilojoules because that's more of a standard value to use for these types of calculations, so keep that in mind. And remember, we can find delta g by using this formula, Δg=Δh−tΔs.
Gibbs Free Energy (Simplified) Example 3
Video transcript
For a particular reaction, ΔH equals -111.4 kilojoules and ΔS equals -25 joules per Kelvin. To calculate ΔG for this reaction at 298 Kelvin, we consider the following equation:
Δ G = Δ H − T Δ SΔH is -111.4 kilojoules, and the temperature (T) is 298 Kelvin. We need to convert our Joules to kilojoules for consistency, so -25 Joules per Kelvin is equivalent to -0.025 kilojoules per Kelvin. Now substituting these values, we get:
Δ G = ( - 111.4 ) − ( 298 × - 0.025 ) k i l o j o u l e sThe calculation yields ΔG as -103.95 kilojoules. Since the ΔG value is less than 0, we can conclude that the reaction is spontaneous at 298 Kelvin. The system is spontaneous as written, i.e., it is not at equilibrium because for equilibrium ΔG would have to equal 0. It would be spontaneous in the reverse direction only if we had gotten a ΔG that is greater than 0, since it would be non-spontaneous in the forward direction and we would have to reverse the reaction to achieve spontaneity.
A particular reaction has ΔG = –350 kJ and ΔS = –350 J/K at 24°C. How much heat will be released/absorbed?
For a reaction in which ΔH = 125 kJ and ΔS = 325 J/K, determine the temperature in Celsius above which the reaction is spontaneous.
Do you want more practice?
Your GOB Chemistry tutor
- The change of state from liquid H2O to gaseous H2O has ∆H = +9.72 kcal/mol(+40.7 kJ/mol) and ∆S = -26.1 cal/(...
- The change of state from liquid H2O to gaseous H2O has ∆H = +9.72 kcal/mol(+40.7 kJ/mol) and ∆S = -26.1 cal/(...
- Two curves are shown in the following energy diagram: Which curve represents the spontaneous reaction, and w...
- The following diagram portrays a reaction of the type A(s) → B(g) + C(g), where the different-colored spheres ...
- The following diagram portrays a reaction of the type A(s) → B(g) + C(g), where the different-colored spheres ...
- What two factors affect the spontaneity of a reaction?
- What is the difference between an exothermic reaction and an exergonic reaction?
- Under what conditions might a reaction be endothermic but exergonic? Explain.
- For the reaction 2 Hg(l) O2 → 2HgO(s), ∆H = -43 kcal/mol (-180 kJ/mol). Under what conditions would you expec...
- The reaction between hydrogen gas and carbon to produce the gas known as ethylene is: 2 H2(g) + 2 C(s) → H2C=...