In these series of videos, we'll learn what ionic hydrates are and the rules needed to name them. Now we're going to say ionic hydrates are simply ionic compounds linked to at least one molecule of water. Here we have as our example CuxH2O. X just represents some unknown number of water molecules that are linked to copper and in chlorine together here. Now the dot here is what's referring to as the linking or bonding of the water molecules to this ionic compound. Now that we know what an ionic hydrate is, let's look at the rules to name them. Some of these rules are pretty familiar to us because a big chunk of it has to do with naming ionic compounds, which we've covered in earlier videos. Now steps 0 to 3 are the same rules as naming ionic compounds. If you don't remember those rules, make sure you go back and take a look at our videos on naming ionic compounds. Step 4 is new. For step 4, we're going to say that the water portion will be called hydrate, but we also have to specify how many of these hydrates are connected to my ionic compound. We're going to say to describe the number of water molecules in a hydrate, we now use numerical prefixes. Now these numerical prefixes go from mono to deca, mono meaning 1, and then di is 2, tri is 3, tetra is 4. Some of these terms we are familiar with. Pentagon has 5 sides, so penta has 5. Hexagon has 6 sides. Hepta, we may not have been familiar with that because that's not a term that you usually see. So hepta is 7. Octagon, 8. Now nona is 9, and deca is 10. So now that we know how to identify ionic hydrates and we've learned the basic rules for naming them, let's take a look at some questions and put what we learned into practice.
Naming Ionic Hydrates - Online Tutor, Practice Problems & Exam Prep
An Ionic Hydrate is an ionic compound that is linked to at least one molecule of H2O.
Naming Ionic Hydrates
Naming Ionic Hydrates
Video transcript
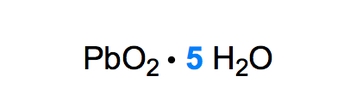
Naming Ionic Hydrates Example 1
Video transcript
In this example question, we're told to name the following ionic hydrate. Alright. So let's do the easy part first. We have 5 water molecules here. So 5 would mean that we're using the numerical prefix of penta, and the water would be called hydrate. So this is pentahydrate. Now, if we take a look at the ionic compound portion, we have lead with oxygen. With lead, we have an issue though. Lead can have a charge of either 2+ or 4+. Remember, lead is not a transition metal, but it and tin have this issue where they can be 2+ or 4+. If you don't remember that, again, go back and take a look at my periodic table videos that deal with charges. Now here, oxygen is in group 6A. And remember, if you're in group 6A, your charge is -2. So let's think about this. Oxygen is -2, there are 2 oxygens within the formula. So collectively, the oxygens together are -4. For PbO2 to be neutral, this -4 has to be counterbalanced or canceled out by what? By a +4. So the +4 of lead cancels out the -4 from the 2 oxygens, thereby giving us a neutral ionic compound. So that would mean that we're dealing with, for our ionic compound, lead(IV), remember, it uses Roman numerals because it has multiple charges, lead(IV) oxide pentahydrate. So this one was tricky, but this would be the name of our ionic hydrate.
Give the systematic name from the given formula:K2Cr2O7 ∙ 3 H2O
Write the chemical formula for the following compound:Calcium selenide hexahydrate
