Now, emission spectra is a series of lines formed when emitted light is focused by a slit and passes through a prism. So remember, we have our atom here, and here we have our first shell. And theoretically, there's an infinite number of shells within a given atom. We haven't found all the elements in the universe, so there are elements out there that we still don't know about. So the number of shells is infinite. And we're going to say here we have an electron that's at a higher energy state and a higher energy level, and what's going to happen is eventually it starts to come back down to its ground state. So it's dropping back down to the first shell. Remember, as an electron drops back down from a higher shell to a lower shell, it emits energy. This energy is emitted as light. What happens here is the slit will focus this energy, and what happens there is it passes through a prism. This prism allows us to split that energy into its various wavelengths and create this emission spectrum. So remember, a slit is just a narrow long narrow cut used to spread closely packed wavelengths which can later be measured. So we can examine this emission spectrum and we can measure the wavelengths of each one of these colored lines. Just realize here that the prism itself, it helps to transform these wavelengths into discrete lines on the emission spectra. So all that's really going on here is we're focusing the emitted energy as an electron drops from a higher shell number down to a lower shell number and imposing them on an emission spectrum. From there, we can calculate their wavelengths, and in that way also calculate the energy or frequency of these different wavelengths of light.
Emission Spectrum (Simplified) - Online Tutor, Practice Problems & Exam Prep
Emission Spectra is a series of lines formed when emitted light is focused by a slit and passed through a prism.
Emission Spectrum
Emission Spectrum (Simplified) Concept 1
Video transcript
Emission Spectrum (Simplified) Example 1
Video transcript
The lines in an atomic emission spectrum are due to the presence of isotopes. Well, when we talked about the emission spectrum that's created, we never mentioned isotopes. Movement of electrons from higher energy states to lower energy states in atoms. We did say this. We said that as the electron falls from a higher energy level or shell number to a lower one, it emits energy in the form of light. The slit focuses this energy through a prism in order to superimpose it on an emission spectrum. So this is true. Here, nuclear transitions in atoms, which we never discussed, and then this is saying the opposite. It's saying we're going from a lower energy state to a higher energy state. This would require absorption of energy, so the slit wouldn't be able to focus any emitted energy to create our emission spectrum. So this is the opposite of what is needed. So here, option b would be the best answer, and remember the name is emission spectrum. So remember what emission is. We're going from a higher energy state or shell number to a lower one. That should have been a key indicator that option b was the best answer.
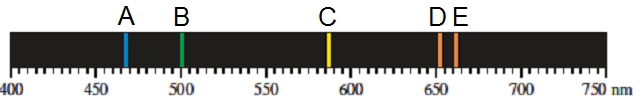
The emission spectrum of helium is shown below. Which emission spectrum line has the highest energy?