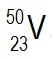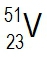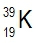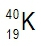Atomic masses of elements can be found by simply looking at the periodic table. So let's start off by looking at the symbol of H, which represents hydrogen. And if you take a look at hydrogen as well as the other elements on the periodic table, you'll see these whole numbers. These whole numbers represent our atomic number. They are the number of protons. When we say atomic mass though, the atomic mass is the number that is seldom a whole number. This is our atomic mass. So you can find the atomic mass of any element on the periodic table just by simply looking it up. Now we're going to say the atomic mass itself is an average of all its isotopes that use the units of grams per mole, AMU, or Daltons, and we're going to say remember that 1 AMU equals 1.66 × 10 - 27 kilograms. So just remember, these atomic masses that you see on the periodic table, they are usually not whole numbers. You'd have to get way down below here to these heavy elements down here till you see whole numbers for atomic masses. And remember, they are the average of all the isotopes for that given element.
Atomic Mass (Conceptual) - Online Tutor, Practice Problems & Exam Prep
The atomic mass of an element can be found on the Periodic Table.
Determining Atomic Mass
Atomic Mass (Conceptual) Concept 1
Video transcript
Atomic Mass (Conceptual) Example 1
Video transcript
So here for this example question, it says, which of the following represents an element from the first column with the greatest atomic mass? Alright. So our first column, if we look at this periodic table, includes all of these different elements. And remember, the number in red, which is not a whole number normally, that represents the atomic mass of any of these given elements. Now here, if we take a look, we have barium, Ba. Again, later we'll learn about how the names are attached to the element symbol. Ba is not in the 1st column; here it's in the 2nd column. So this cannot be a choice. Then we're going to say next that we have Al. Al stands for aluminum. Aluminum is over here in the 3rd column, well, all the way over here in this 13th column, actually. So, this is out. Next, we have Cs, which is cesium. Here it is right here. It's in the first column. It's pretty low down there. It's 132.91 for its atomic mass. Remember, that could be in grams per mole, atomic mass units, or Daltons. So far, it looks like it's the highest one. The only one higher than that would be Fr. Notice that in the bottom rows here, most of them are whole numbers. These are super large mass elements that are pretty unstable. They typically don't have numerous isotopes. As a result, they have no decimal places. So, so far C looks like it's our best choice. If we look at D, we have Li, which is up here, not higher in mass, not greater in atomic mass. And then we have Na, which is right here. So it looks like C is our best choice. It has the greatest mass, atomic mass, from column 1 from the choices provided. So just remember we have our element symbols, we have our atomic masses, which normally are not whole numbers, and then we actually have whole numbers. Those represent our atomic numbers.
On the Periodic Table, the atomic mass is represented by the number with decimal places.
Which of the following choices has the greatest atomic mass?
Atomic Mass (Conceptual) Concept 2
Video transcript
When dealing with isotopes, we can talk about the most abundant isotope. Recall, the atomic mass of an element is an average mass of all its isotopes, and we're going to say that the most abundant isotope for an element is the one with a mass number closest to the atomic mass of the element. Remember, your mass number gives you the number of protons and neutrons together for a given isotope. So if we take a look here, we have our element, their atomic mass, the isotope symbols, and then we talk about their most abundant isotope. So for hydrogen, its atomic mass, according to the periodic table, is 1.008 amu and its isotope symbols are hydrogen. When it has a mass number of 2 it's called deuterium, and when it has a mass number of 3, it's called tritium. Alright. Now, remember, we said that the most abundant isotope is the one that has a mass number closest to the atomic mass. The atomic mass is 1. Here are our mass numbers, 1, 2, and 3. The one closest to the atomic mass is hydrogen-1. Its atomic mass number is 1 and it's closest to the atomic mass of 1.008.
Let's go to the next one, boron. Boron has two forms, boron-10 and boron-11. The atomic mass of boron is 10.81 amu. Looking at boron-10 and boron-11, which one has a mass number closest to this atomic mass? The answer would be boron-11 because 11 is closer to 10.81 than 10 is.
Then finally, we have sulfur. Sulfur has an atomic mass of 32.06 amu when you look on the periodic table. Alright? Now, some versions might show 32.07, but then remember, it's okay. It can either be 32.06 or 32.07, depending on which periodic table you are looking at. But that doesn't matter. Here is my atomic mass. Which mass number is closest to that 32.06? And we see that the answer is sulfur-32. 32 is closer to 32.06 than either 33, 34, or 36. Alright. So just remember, the most abundant isotope, to figure it out, look at what the atomic mass of the element is on the periodic table, look at the different mass numbers for all the isotopes, the one that has a mass number closest to that atomic mass is the most abundant isotope.
Atomic Mass (Conceptual) Example 2
Video transcript
Oxygen consists of 3 isotopes, oxygen-16, oxygen-17, and oxygen-18. If the atomic mass for oxygen on the periodic table is 15.999 AMU, which isotope is the most abundant? So remember, we look at the atomic mass, which you can find on the periodic table for the element, and then you look to see which mass number is closest to it. The one closest to it represents the most abundant isotope. Oxygen-16 would have to be the answer because its number of 16 is closest to the atomic mass of 15.999 AMU.
Vanadium consists of two isotopes, 5023V and 5123V. If the atomic mass for copper on the periodic table is 50.942 amu, are there more atoms of 5023V and 5123V in a sample of vanadium?
Potassium consists of three isotopes, 3919K, 4019K, and 4119K. Based on its atomic mass, which isotope of potassium is the most abundant?