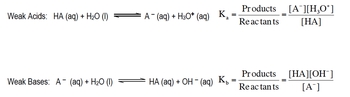
Hey, guys. In this new video, we're going to take a look at the acid and base constants. So, as you guys can know from looking at the rules for identifying compounds as either acids or bases, you should realize that there's a lot more weak species out there than there are strong ones. What we need to realize is that these weak species have constants associated with them. We're going to say here, consider a weak monoprotic acid. Monoprotic just means it has 1 H positive. And here we're looking at how it ionizes in water. Basically, is the acid so it's going to give away an H positive to the water. The water accepts the H positive so that's how it becomes H3O+ The acid loses an H positive and that's how it becomes A-. Now what we're going to say here, the equilibrium expression equals products over reactants. And remember, we've talked about the equilibrium expression before. It looks at all phases of matter except for liquids and solids. So liquids and solids will not be included. And here, we have a liquid, so we're going to ignore it when we do the equilibrium expression. It's simply going to become A-∙H3O+ over
Ka and Kb - Online Tutor, Practice Problems & Exam Prep
Associated with any weak acid or weak base is a Ka or Kb value respectively.
Acid and Base Dissociation Constants.
Ka and Kb Concept 1
Video transcript
Weak acids possess a Ka value less than 1, while weak bases possess a Kb value less than 1. The equilibrium expressions of Ka and Kb are the same as other equilibrium constants we've seen.
Ka and Kb Concept 2
Video transcript
Now we're going to say that \( K_a \) represents our acid dissociation constant. And it basically measures how strong our weak acids are. We're going to say weak acids use \( K_a \), weak bases don't use \( K_a \). Instead, they use \( K_b \). So this is our base dissociation constant. And what we're going to say here is their relationship to each other \( K_a \) and \( K_b \) can be expressed by this formula. \( K_w = K_a \times K_b \). And as you realize, this \( K_w \) is the same \( K_w \) we saw earlier. In the earlier times, we saw it as \( K_w = [\text{H}^+] \times [\text{OH}^-] \). It's the same exact \( K_w \). And we're going to say in general, the higher the \( K_a \) value and the stronger the acid. The stronger the acid, the greater the concentration of \( [\text{H}^+] \). Now we're going to say related to \( K_a \) is \( pK_a \). And remember, we've talked about what \( p \) means. \( P \) means negative log. \( pK_a \) means negative log of \( K_a \). Here we're going to say there's an inverse relationship. It's actually the lower the \( pK_a \), the stronger the acid and the stronger the acid, the greater the \( [\text{H}^+] \) concentration. Now, we're going to say here is when it comes to a weak acid, we're going to say weak acid tends to have \( K_a \) value less than 1 and a \( pK_a \) value greater than 1. Just realize we said inverse relationship, so \( K_a \) and \( pK_a \) are opposites. If one is up, the other one is down. Strong acids though, strong acids tend to have \( K_a \)'s much greater than 1. And they tend to have \( pK_a \)'s lower than 1, \( pK_a \)'s that are negative. And remember, we should be able to establish a relationship between \( K_a \) and \( pK_a \) in the same way we've been able to establish a relationship between \( [\text{H}^+] \) and \( pH \). Because here, we can just simply say \( K_a = 10^{-pK_a} \). We could also say the same thing for \( pK_b \). \( pK_b = \text{negative log of } K_b \) and therefore, \( K_b = 10^{-pK_b} \). Just remember these relationships and how they affect the acidity of our compound. As long as you can remember these few quick easy calculations, you'll be able to go from one to the other. And one last thing, we can say here that the relationship from \( p \) to \( pK_a \) and \( pK_b \) can be established by looking at this equation here. By taking the negative log of \( K_w \), \( K_a \) and \( K_b \), we say that the equation becomes \( 14 = pK_a + pK_b \). Now, that we've seen all these types of new variables and new types of equations, attempt to do the practice question on the bottom. Here I say if the \( K_b \) of \( \text{NH}_3 \) is \( 1.76 \times 10^{-5} \), what is the acid dissociation constant of the conjugate acid? Conjugate acid is a term we learned earlier a few videos back. But here we're given \( K_b \), we're asked to find the acid dissociation constant. So it's a simple conversion from one unit to another. So, once you're done with that, click back on the explanation button and see how I approach this problem. Good luck, guys.
The greater the Ka value then the stronger the acid, while the greater the Kb the stronger the base. Ka and Kb are connected by the following equation:
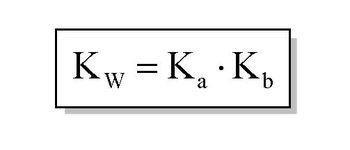
Ka and Kb Example 1
Video transcript
Hey, guys. In this new video, we're going to put to practice some of the concepts we learned about Ka and Kb. So the first example says, knowing that HF has a larger or higher Ka value than CH3COOH which is acetic acid, determine if possible in which direction the following equilibrium lies. We learned earlier that the higher your Ka value, the stronger you are as an acid. HF has a higher Ka value, so it's a stronger acid than acetic acid is. We're going to say HF is the stronger acid. And again, it's stronger because it has a higher Ka value. And it's the stronger acid, so the acetic acid on the product side would be the weaker acid. Now, we have to say on which side does the equilibrium lie. All you have to realize here is weaker means more stable. In chemistry, weaker just means less energy. Less energy means you're more stable. And the equilibrium will always lie on the side with the weaker acid because the weaker acid is the more stable acid. Our weaker acid is on the right side here. We're going to say that equilibrium lies to the right. It's as simple as that. First, you have to know that higher Ka means stronger acid and then just figure out which side the weaker acid would be on. Wherever the weaker acid is, that's where your equilibrium will lie.
If the Kb of NH3 is 1.76 x 10-5, determine the acid dissociation constant of its conjugate acid.
Which Bronsted-Lowry base has the greatest concentration of hydroxide ions?
Which Bronsted-Lowry acid has the weakest conjugate base?
