In this video, we'll focus on amino acid catabolism in relation to the amino group. Now, here we're going to say the first stage of amino acid catabolism is the removal of the amino group in the liver. Now, this is a two-step process. We're going to say process A is called transamination, process B or process 2 is oxidative deamination. Now, if we take a look here, we see that we have our macromolecules of proteins, carbohydrates, and lipids. You're paying close attention to the digestion of proteins, so we're seeing how that creates our amino acids. Within our cytosol here, we're going to see that our amino acids through transamination, we have the removal of the amino group, and that occurs within the cytosol. Here, if we take a look, our amino acid, we have transamination. We see that we can have the formation of what we call an alpha-keto acid. We'll go into greater details of this; this is just a broad overview of what's going on. We're going to say here, through transamination we also have the generation of what's called Glutamate. This occurs within the mitochondria. And here, we are looking at the mitochondrial matrix in which it occurs. Now, here, once we have our Glutamate, we go into the second part of this process which is oxidative deamination. This is the formation of our Ammonium ion which is NH4+ and again it's occurring within the mitochondria. So through oxidative deamination, we have the formation of the ammonium ion. And then, we also have if we cross the mitochondrial membrane back into the cytosol, we have the alpha-ketoglutarate being produced as well. So here we have stage 1, stage 2 is occurring within the cytosol. We're going to say here that part of this traverses across the mitochondrial membrane into the mitochondrial matrix where we have glutamate, the production of the ammonium ion, and then going back across the mitochondrial membrane, we have our Alpha Ketoglutarate. So just keep in mind, when it comes to amino acid catabolism in relation to the amino group, we have transamination and then oxidative deamination.
Amino Acid Catabolism: Amino Group - Online Tutor, Practice Problems & Exam Prep
Amino Acid Catabolism: Amino Group Concept 1
Video transcript
Amino Acid Catabolism: Amino Group Concept 2
Video transcript
Hey, everyone. So in this video, let's talk about transamination. Now first, we're going to say that it is a reversible reaction. And we're going to say here in transamination, we're going to say the amino group of the amino acid and the Keto group of the Alpha Keto acid are exchanged. If we take a look here at our reactants, remember this is our amino acid because it has the four parts that make it an amino acid. We have our carboxyl group. We have our amino group, we have our alpha hydrogen here, and then we have our R group. Here, what makes this an Alpha Keto Acid? Well, here it's an acid because it has this carboxyl group that can be acidic. And then remember, Alpha, this carbonyl, the C double bond O that's part of this structure that I circled, that is our main carbonyl. The carbon next to it is our Alpha Carbon. So this is our Alpha Carbon. That Alpha Carbon is a carbonyl as well. That's why we call it an Alpha Keto because it's a Keto. And then we have our R group. Now, the exchanging between the Amino group and the Keto group is accomplished through the use of our Enzyme. That enzyme is transaminase. You might also hear it being called Aminotransferase. Okay. So either term could be used for it. So, again we said that this is an exchange. So all that's happening is this group and this, they're exchanging. They're swapping. What's the result of this? Well, here we have our new carbonyl here to create our new alpha, keto acid, and then we have our new our ammonia, amino group being put here to create our new amino acid. Now what's the takeaway from here? We're going to say the alpha keto acid is usually an alpha ketoglutarate, and that produces Glutamate as the new amino acid. So that's the key takeaway from transamination. We have the exchanging between an amino group and a keto group.
Amino Acid Catabolism: Amino Group Example 1
Video transcript
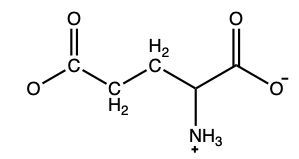
In this example, it says complete a transamination reaction for aspartate. Remember what happens here is we have the exchanging between an amino group of the amino acid and the keto group of an alpha keto acid. If we take a look here, here is our amino group of this amino acid and here is our alpha carbonyl group of this alpha keto acid. Remember, it is the double bond O that's exchanging. Right? So they're kind of swapping places. So what effect would this have? Well, here we have our reversible arrows because remember this process is reversible. We would show this first one. We'd still draw it. So we still have this portion with the H up here, CH2, and then this. Remember we're swapping. So now this, oops. And actually, this carbon needs to make 4 bonds so it loses that hydrogen too. We'd have our double bond and then O here. So this is our new group being formed. Plus, we're going to say we have this other one now is transformed. We'd have our NH3+ here. Carbon still needs to make 4 bonds so it'd also have an H here as well. So this would represent our 2 products being formed through transamination when we're dealing with aspartate.
Amino Acid Catabolism: Amino Group Concept 3
Video transcript
In our continued discussion of amino acid catabolism in relation to the amino group, we're now going to look at the second step or phase b of our process. Here, we're talking about oxidative deamination. Now, here we're going to say that the glutamate is going to be oxidized back to an alpha-ketoglutarate by NAD+. This is going to help form our ammonium ion. The ammonium ion itself then enters the urea cycle. If we take a look here, we have our glutamate as a reactant, plus water. We're utilizing our enzyme glutamate dehydrogenase. Remember, naming the enzyme is easy. It's the name of the substrate, which happens to be glutamate. Because this is an oxidation, remember that we utilize the class of enzymes called dehydrogenase. Here, we have NAD+. NAD+ is reduced to NADH, a high-energy molecule. NAD+ being reduced is what causes glutamate to become oxidized. So what's going to happen here is we're going to transform it into an alpha-ketoglutarate. This carbon now is going to have the carbonyl. And remember we're also going to produce our ammonium ion. These will represent the products formed within this oxidative deamination and they would be our final answer.
Amino Acid Catabolism: Amino Group Example 2
Video transcript
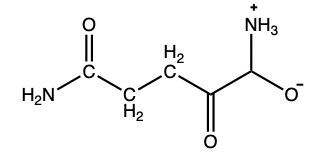
In this example question, it says complete a transaminase and oxidative deamination reactions for threonine. Now, here's the thing, threonine is just one of those unique amino acids, as well as proline and lysine that don't undergo transamination. Proline and lysine don't undergo transamination because they lack the alpha amino group. Threonine, however, doesn't undergo it because it would create a dimerized toxic type of structure. But let's just forego that limitation and imagine that if it were to undergo transamination, what would happen? Now remember, in transamination, the way it would work is we'd say that this serves as our amino acid. We'd have to react it with an alpha ketoacid, and here's our alpha ketoacid. Remember, with this alpha ketoacid through transamination, we would basically exchange the amino group and the keto group. This would change this amino acid into this new alpha ketoacid, and we make a new amino acid here. So this new amino acid would look like this. Remember, this new amino acid that we make is Glutamate. Glutamate itself would then undergo oxidative deamination. So here we're going to break it down. Here, since this is going to represent Glutamate, we're going to write in what this R group represents. So, for Glutamate, it would be our carboxyl group. And then we'd have our CH2 groups. We have our CH here. Here's the amino group that was added. Here's our other carboxyl group. Remember, we react it with water as well. We'd use NAD+, it becomes NADH. We'd use glutamate dehydrogenase in order to do this oxidation. So all that happens here, here is still our carboxyl group. And this NH3, this amino group is transformed into a carbonyl. This would help to create our alpha keto butyrate. This would be our final product here.
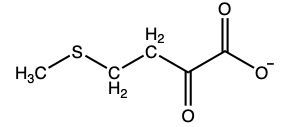
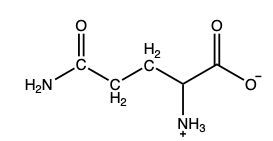
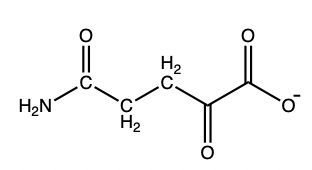
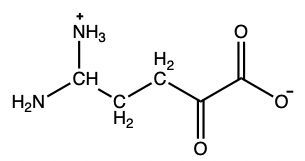
What amino acid yields the following α-keto acid through transamination?
Lysine
Methionine
Cysteine
Glutamate
Draw α-keto acid produced by transamination of glutamine.
Do you want more practice?
Your GOB Chemistry tutor
- Unlike most amino acids, branched-chain amino acids are broken down in tissues other than the liver. Using Tab...
- Write the structure of the α-keto acid produced by oxidative deamination of the following amino acids (Refer t...
- What other product is formed in oxidative deamination besides an α-keto acid?
- Draw the condensed structural formulas for the products of the reaction of aspartate and α-ketoglutarate which...