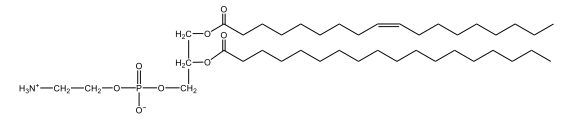
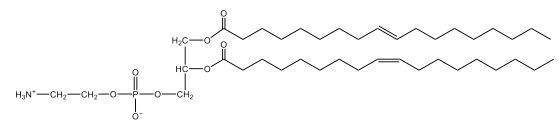
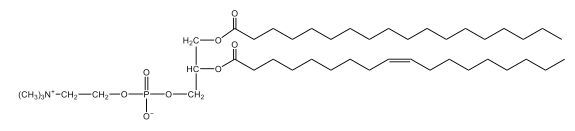
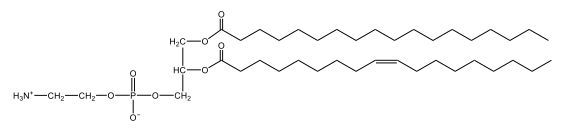
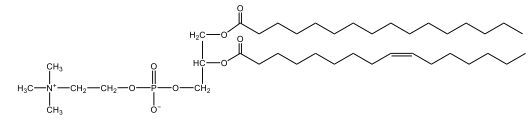
Now, here we're going to say that phospholipids are lipids that contain a phosphate group attached to a glycerol or sphingosine backbone. Here we're going to say, like fatty acids, phospholipids are amphipathic, meaning they have a hydrophilic head and a hydrophobic tail. If we take a look here, we have our illustration of our hydrophilic head. Because it's hydrophilic, it would be a polar head. And then we have our hydrophobic tail, which will mean it's non-polar. Now remember, lipids can be broken down first into fatty acids and steroids. Here we're focusing on the fatty acid portion, which can be further broken down into our waxes, our glycerol lipids, our sphingolipids, and then here we'll talk about our eicosanoids later on. And here, when we're talking about our phospholipids, it's actually shared by these two subclassifications here. Because both of them contain a phosphate group attached to a glycerol or a sphingosine backbone. Now, if we understand that our head is polar and hydrophilic, and our tail is hydrophobic, and this was to be submerged in an aqueous environment, it would orient itself to create this lipid bilayer, where the polar heads are on the exterior and the hydrophobic tails are in the interior. Now, this is important because its ability to do this means that phospholipids are a major component of all cell membranes. Right. So that's an important thing we need to take into account when dealing with phospholipids within aqueous or polar environments. Right? So just keep this in mind in terms of the classifications of phospholipids.
Glycerophospholipids - Online Tutor, Practice Problems & Exam Prep
Glycerophospholipids Concept 1
Video transcript
Glycerophospholipids Example 1
Video transcript
Which one of the following is not a component of phospholipids? So we did talk about phospholipids being involved with fatty acids because it is underneath the umbrella term of fatty acids. Phosphate. We said that phospholipids, a big important aspect of them, is that they contain a phosphate group. It can be connected to a glycerol molecule or a sphingosine molecule. Cholesterol. Cholesterol here is not a component of phospholipids, and in fact, it belongs under the category of steroids. So here, this would be our answer. Then finally, Glycerol. Glycerol can be a vital component of phospholipids. It forms a potential backbone in terms of the attachment of the phosphate group. So this would also be a component of phospholipids. So here the only answer that's correct is option c. Cholesterol is not a component of phospholipids.
Glycerophospholipids Concept 2
Video transcript
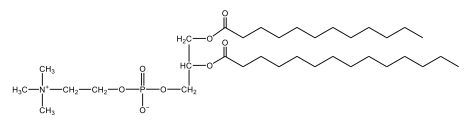
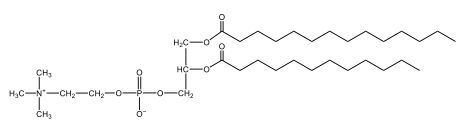
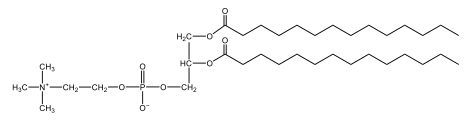
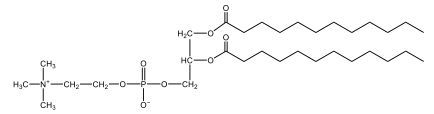
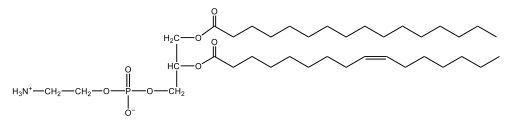
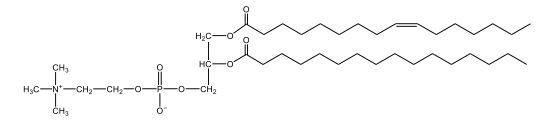
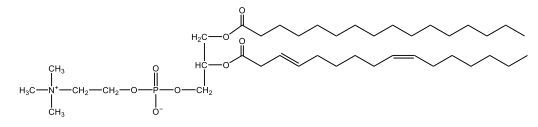
Now, Glycerophospholipids are also called phosphoglycerides, and they are phospholipids with a glycerol backbone and 2 fatty acids. We're going to say here, it has a head and tails, and we're going to say the head portion is a phosphate group extended with an amino alcohol head group. The tail is just 2 fatty acids attached through ester bonds. Now this is important, they're classified based on the head group attached to the phosphate group. If we take a look here, we have two classes that exist. We have Cefalins and then we have Lecithins. Here, when it comes to Cefalins, the head group is an ethanolamine. Here the nitrogen is connected to 3 hydrogens, an ethyl and then followed by an alcohol. This alcohol portion is what bonds to the phosphate group, so, this is the head region. The fatty acid chains could be saturated or unsaturated, again that's not part of the classification. It's the presence of the Ethanolamine group that makes it a Cefalin. Now, Lecithin, here we're going to have choline as our head group. And the clue here, choline CH, instead of having hydrogens that we have methyl groups. And we still have our ethyl and then an OH. That OH is reacting with a phosphate group for the attachment. Now, here we're going to say what's important here and also the fatty acid chains could be saturated or unsaturated. Now, here when it comes to our Glycerophospholipids, they are the most abundant lipids in cell membranes. But remember, the classification of these types of Glycerophospholipids is based on the type of head group.
Glycerophospholipids Example 2
Video transcript
This example question asks, "What is the basis of the classification of Glycerophospholipids?" Now remember, we said that it is the type of head group that's attached to our phosphate that determines the classification for Glycerophospholipids. So we take a look here; it is not about the fatty acid molecule of carbon 1, the number of double bonds in carbon 2 fatty acid, or the fatty acid molecule of carbon 2. Again, it is the head group that's attached to the phosphate group. And remember, here we can have either an ethanolamine head group or a choline head group as the Head Group. In both cases, we have a nitrogen making 4 bonds and therefore it's positively charged. With an ethanolamine head group, we have nitrogen connected to 3 hydrogens, an ethyl group then connected to an alcohol. And then for choline, the nitrogen is not connected to hydrogens but to 3 methyl groups and it still possesses a positive charge. So, in this particular question, it is the head group attached to the phosphate group that deals with the classification of our Glycerophospholipids.
Which one of the following statements accurately describes the difference between cephalins and lecithins?
Cephalins contain saturated fatty acids while lecithins have unsaturated fatty acids.
Lecithins and cephalins have different backbone molecules.
The head groups in cephalins and lecithins are ethanolamine and choline, respectively.
Lecithins do not have a head group.
Glycerophospholipids Example 3
Video transcript
Now, when it comes to drawing Glycerophospholipids, we're going to say it requires recalling the structures of the fatty acids and head groups. Alright. So here it says to draw the structure of a Glycerophospholipid that contains 2 lauric acid acetyl groups and ethanol amine bonded to the phosphate group. Alright. So here, the way we're going to approach this is step 1, we're going to draw the Glycerol backbone with the phosphate group at carbon number 3. So remember, we're going to say this is 123, we're going to say instead of 2 OH groups at carbon 1 and 2, we're going to write only oxygen atoms. So here, carbon 1 and 2 we have an oxygen here and an oxygen here, we already have our phosphate group connected to Carbon 3. Next, we're going to say step 2, extend the Phosphate group at Carbon 3 with, CH 2CH 2 group and an ethyl group. And we're going to say we're going to complete the head group with a NH3+ group because it's ethanolamine, or we're going to complete it with a nitrogen connected to 3 methyl groups, and it's also positive if it's a choline group. So here we're going to say we have CHCH 2 attached. They told us it's an ethanolamine, so it's going to be an NH3+ group attached. And then let's see. So then we're done with that portion. And then finally, it says draw the 2 fatty acyl groups. So here we're going to say, remember, we don't include the OH group of the fatty acid from the 2 oxygen atoms at carbons 1 and 2. Alright. So we have our ethanolamine group attached, and they're telling us within this question it's 2 lauric acid acetyl groups. Remember, lauric acid is going to be a saturated fatty acid. It has 12 carbons in total and it has no pi bonds. So we just have to draw that out. So 2, 4, 6, 8, 10, 12. Same thing here. 2, 4, 6, 8, 10, 12. So here, this would be the structure of our Glycerophospholipid based on the description at the beginning of the example question.
Draw a glycerophospholipid with lauric acid at C1, myristic acid at C2, and choline bonded to phosphate.
Draw a cephalin with stearic acid at C1 and oleic acid at C2.
Draw a lecithin with palmitic acid at C1 and palmitoleic acid at C2.