Now, coenzymes are important in metabolic reactions. We're going to say the driving force of catabolism is the oxidation of molecules in order to release energy. We're going to say this is accomplished by coenzyme cycling between their oxidized and reduced forms. Now, here we're going to say the reduced forms act as electron carriers that carry energy that is ultimately passed to the bonds of ATP. If we take a look here, we're going to say that we have substrate A. And to show its reduced form and remember, reduction here is in terms of having hydrogen or not, gaining hydrogen or not. So this substrate A would be the reduced form of it. To show that, we just show a hydrogen on it. Here it undergoes oxidation where it will lose its hydrogen. So if this thing here is losing its hydrogen, where is it going? Well, if it's losing its H, it's handing it over to the coenzyme. Here, the coenzyme in its oxidized form doesn't have it, but it gets reduced in this process and now it's in its reduced form. The coenzyme is the carrier of the electrons in the form of this hydrogen component. That hydrogen is not only H, it's the electrons in the bond with it. That reduced form can then take itself, become oxidized back to its oxidized form. When it becomes oxidized, it's basically handing over its H to substrate B. Substrate B gains that H, and now we have it in its reduced form here. So, basically, these coenzymes are kind of acting as a carrier. They're taking electrons from one molecule and handing them over to another one later on. This, again, ultimately is to help us to create lots and lots of ATP at the end. So as we progress through our topics, we'll see how this exactly is done.
Coenzymes in Metabolism - Online Tutor, Practice Problems & Exam Prep
Coenzymes in Metabolism Concept 1
Video transcript
Coenzymes in Metabolism Example 1
Video transcript
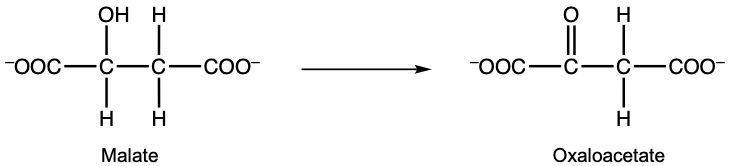
Here in this example, it says, consider the reaction given below and correctly identify the oxidizing agent. Remember, your oxidizing agent is what's been reduced. And what's been reduced has gained electrons or, in this case, Hydrogen with those electrons. And remember, when we're looking at oxidizing agent or reducing agent, those are the reactants. We look at the product side to see where the electrons went, but then we look back at the reactant to determine, okay, this was reduced, and this was oxidized. If we take a look here, we have malate as a reactant, and then over here, we have Oxaloacetate. Here we have NAD+ which is a coenzyme. But if we look, NAD+ in this form as a reactant, but then over here, it's gained a hydrogen. And not only a Hydrogen but electrons. That's why it's neutral now. So we're going to say NAD+ gained electrons and Hydrogen and so it was reduced, and therefore it's the oxidizing agent. Now this question doesn't ask this, but we could also say that Malate had an alcohol group here. This alcohol group was oxidized into a ketone portion here. Remember, we learned this several chapters ago that alcohols can be oxidized into carbonyl groups C=O. So this thing was oxidized. Therefore, it's the reducing agent. The question doesn't ask this, but we're just trying to cover everything to see the difference. But again, for this particular one, the oxidizing agent would be NAD+.
Nicotinamide Adenine Dinucleotide Concept 2
Video transcript
So, Nicotinamide Adenine Dinucleotide, or NAD. We're going to say here the Nicotinamide group of NAD+ is the site of reduction that is seeking to become neutral, so we want to lose that charge. If we look here at NAD+, it's oxidized form. Nitrogen, remember when it makes 4 bonds, is positively charged. We're trying to remove this positive charge here. We're going to say the reduction occurs by accepting 2 electrons. We have 2 electrons here. And, we're going to say here to gain 1 Hydrogen. So here, we're going to gain the hydrogen, the hydrogen will be gained right here. And, initially, we're going to start with 2 Hydrogens. We used 1 to attach it to our structure to create NADH, the reduced form, so it'd be one left.
Now, here are the results of the reduction of NAD+ to NADH. If we take a look here, the way this works is that both of these H+s have no electrons whatsoever. And we're going to say here that 2 of these electrons would have bound to 1 of these H+s. So 2 electrons plus the 1 H+ would give us an H-. Gain those 2 electrons. It's this H- that is attaching itself to this site right here. By attaching it to that site there, that's how it's connected to it. And attaching there causes a shifting of our pi bonds. That's why the structure now looks like this: Nitrogen here is no longer making 4 bonds so it's no longer positively charged. It has a lone pair on it though. Again, it's not important to see how these pi bonds move around through resonance. What's important to understand here is that we're going to have this being the site where the H- attaches. So it's important to know you have NAD+ plus 2 electrons plus 2 H+ giving us this structure. Being able to show the structure, and that's the reduced form. And then having 1 H+ left over as a product. Right? So that's what we should take away from this particular example reaction.
Nicotinamide Adenine Dinucleotide Example 2
Video transcript
Which of the following correctly identifies the molecules in the given reaction? So here we have lactate interacting with our coenzyme. Our enzyme is lactate dehydrogenase, and it's going to create pyruvate plus NADH plus H+. Now remember, NAD+ represents a coenzyme and it's in its oxidized form. If we look at our options, b would be out because NAD+ is not in its reduced form. Next, let's see. We can say that lactate dehydrogenase represents our enzyme. We can say here that this lactate represents our substrate. What kind of substrate what form is it in here? Well, here this is the oxidized form, and over here is its reduced form. So we'd say that NAD+ was reduced. Right? To give us this. Right now, it's in its oxidized form. If this is in its oxidized form and this is a redox reaction, the substrate would have to be in its reduced form. And we know it's in its reduced form because its OH group is later on oxidized to a carbonyl group. So the substrate here is in its reduced form. So, all of those are fine. Let's see. Then we're going to say, NAD+ is an oxidized coenzyme. Oxidized cofactor is just the umbrella term. Remember, cofactors can be either organic or inorganic. Pyruvate. What does pyruvate represent? Well, here this is the reduced substrate, so this would have to be the oxidized substrate. So this is good, this is good, and this is good. Now, it looks like option c or d could be an answer, but what's the best answer? Cofactor is just an umbrella term. We got to be more specific. NAD+ represents an organic cofactor. Therefore, it is a coenzyme. So option d would be the best answer here because it goes one step further in describing what NAD+ is. Yes. It's oxidized, but more specifically, it is an organic cofactor called a coenzyme. So option d would be our final answer.
Flavin Adenine Dinucleotide Concept 3
Video transcript
So FAD is called Flavin Adenine Dinucleotide. And we're going to say the flavin group of FAD is the site of reduction that has 2 Hydrogen atoms added to its Nitrogen atoms. Now, here we're going to say the reduction occurs by adding 2 electrons to 2 H+ in order to form 2 new covalent bonds. The result of this is the reduction of FAD to FADH2. So here we have our oxidized form of the flavin portion of FAD. We're reacting 2 electrons and 2 H+ here. And when we reduce this FAD, we get FADH2. The 2 Hydrogens that we gain, 1 is added here and 1 is added here. Notice that there is a shifting of our pi bonds in order to do this. What's most important is to understand that reduction is happening at this nitrogen and this nitrogen and we have the transformation of FAD to FADH2.
Flavin Adenine Dinucleotide Example 3
Video transcript
Here with this example, it states when FAD oxidizes the substrate, it converts the carbon single carbon bonds to carbon double bonded carbon bonds. Complete the following reaction. Alright. So here we have succinate. We have these 2 CH2 carbons in the middle, and then we have these carboxyl groups at the end. We're basically going to use FAD to go from these single bonded carbon carbon bonds to double bonded carbons. So, now those 2 carbons are double bonded. 4 bonds. This Carbon here we see making 3, so it would only have 1 Hydrogen on it. And then the same thing with this one. Here, I just chose to put them on opposite sides of each other. Doing this would go from succinate to what we call fumarate. Now, it's not important that we know what the name of this molecule is for this question. I'm just naming it for you. Now, where did those 2 Hydrogens go? Well, we see that Succinate lost Hydrogens, so it was oxidized, which would mean that FAD had to be reduced. And we know that when we reduce FAD, it becomes FADH2. So these will be our 2 products formed within this given reaction.
Coenzyme A Concept 4
Video transcript
Now, here we're going to say that coenzyme A is a coenzyme of synthase and it has a high energy thiol bond, so s h bond. It carries an Acetyl group to the Krebs Cycle for energy production by oxidation. If we take a look here, this whole structure represents Coenzyme A. Coenzyme A is made up of 3 portions. We have our ADP portion right here, which is Adenosine Diphosphate. Then we have here pantothenic acid. Pantothenic acid, another common name for it is vitamin B5. So that would be this portion right here. And then, finally, we have our Aminoethanethiol portion, which is this nitrogen portion with these 2 CH2s and then the SH at the end. Again, it's the SH group that has a high energy bond. That hydrogen can be removed from this structure. So just remember, Coenzyme A is just yet another simple type of coenzyme that exists.
Coenzyme A Example 4
Video transcript
Now, which of the following statements correctly describes Coenzyme A? Here we have to select all that apply:
- First, when an Acetyl group is released from Acetyl S CoA, it produces here just our CoA with our thiol group. So, we are removing this Acetyl group here and we replace it with a hydrogen. This is true.
- Vitamin B is present in the active site of our Coenzyme A. No, that's not the active site; it's the Aminoethanethiol portion, the portion that has the SH group that has the high energy bond.
- The primary role of CoA is to oxidize fatty acids. No, that's not its primary role. Its primary role is to carry an Acetyl group to the Krebs Cycle for it to become oxidized and generate energy.
- Coenzyme A is composed of Pantothenic acid, which is vitamin B5, Aminoethanol, and ADP. Yes. These are the three major components of our Coenzyme A.
So here, the only two answers that are true are options A and D.
Select the correct statement.
When FAD is reduced, it gains 2 hydrogen ions and 2 electrons, forming FADH.
NAD+ represents an oxidized form of the coenzyme, and acts as an oxidizing agent.
FADH represents an oxidized form of the coenzyme, and acts as a reducing agent.
After CoA is oxidized, it forms Acetyl CoA.
Is the following reaction an oxidation or reduction? Which coenzyme would be carrying this out, NAD+ or NADH?
oxidation, NAD+
oxidation, NADH
reduction, NAD+
reduction, NADH