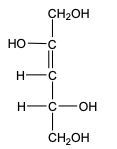
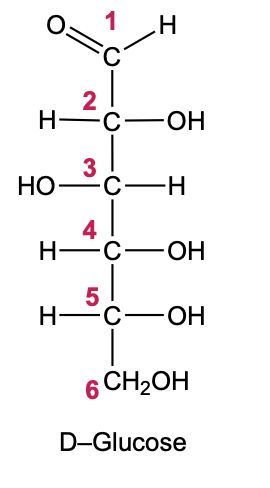
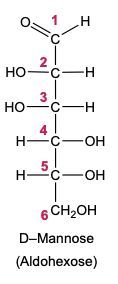
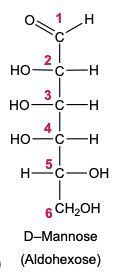
Now when it comes to the reduction of an aldose or keto sugar, we're going to say the carbonyl group is reduced via a reducing agent to a hydroxyl group to create a sugar alcohol. Now when I say hydroxyl group, I just mean an OH group. A sugar alcohol is just a monosaccharide that has all carbons connected to a hydroxyl group. Now when it comes to this reduction, the reducing agent is H2, and the reduction is facilitated by the use of a metal catalyst in the form of nickel, platinum, or palladium. What happens in this reaction is that our carbonyl oxygen gains an H, and the carbonyl carbon also gains an H. So we go from an aldose or keto sugar to our sugar alcohol.
Reduction of Monosaccharides - Online Tutor, Practice Problems & Exam Prep
Reduction of Monosaccharides Concept 1
Video transcript
Reduction of Monosaccharides Concept 2
Video transcript
So, let's look and see what this reduction of monosaccharides would resemble. If we take a look here, we have an Aldo sugar to begin with, and we're going to say we're reducing it with H2 with one of our metal catalysts, nickel, palladium, or platinum. Remember, the carbonyl carbon will gain an H, and this carbonyl oxygen is still present, and it also gains an H. And in that way, we've gone from an Aldo sugar to just a sugar alcohol. Now, let's look at our keto sugar. Same thing. We're using H2 with one of our three metal catalysts. We're going to say here that our carbonyl carbon gains an H, and then the carbonyl oxygen is still present, it also gains an H. And again, we've created a sugar alcohol, this time from a ketose sugar. So remember, we're making sugar alcohols because now all the carbons within this structure have an OH connected to it. Right? So just keep that in mind. We're going from an Aldo sugar to a keto sugar. Our carbonyl group is being transformed into a COH group.
Reduction of Monosaccharides Example 1
Video transcript
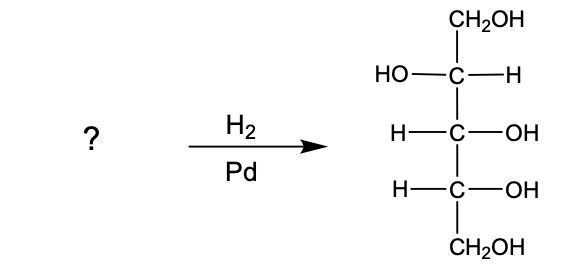
Here it says determine the sugar alcohol product formed from the reduction of the following monosaccharide. So here's our monosaccharide, which is a keto sugar. Remember, in its reduction, it's the carbonyl carbon or the carbonyl group that's being transformed. We're going to say here that that carbonyl carbon gains an H, and the carbonyl oxygen is still there, it also gains an H. And then the rest of the structure stays the same. So we'd still have this C with an H here and an OH here, this C with an OH here and an H here, and then our CH2OH group. So this would be our sugar alcohol product formed from the reduction of this ketose sugar.
Determine which aldose reactant should be used to produce the following sugar alcohol.
Common Naming of Sugar Alcohols Concept 3
Video transcript
We know the reduction of an aldose or keto sugar creates a sugar alcohol. With this, we have to be able to name these sugar alcohols. Now we're going to say the common naming of sugar alcohols is that the set of rules for naming them are similar to aldose and keto sugars. The way we do it is we modify the ending of our aldose or keto sugars, which typically end with "os", and we change it to "-itol." All because it's an alcohol. So remember, when we have an aldose or keto sugar, their ending is "os". When they get reduced to a sugar alcohol, the ending transforms to "itol".
Reduction of Monosaccharides Example 2
Video transcript
Here it says, "Provide the structure and common name for the sugar alcohol created from the reduction reaction." So, we're starting out with D-ribose. We know that when it comes to reduction, we're reducing the aldehyde group. Now remember the aldehyde group is C double bond O connected to an H. When it comes to reduction, the carbonyl carbon would gain an H and the carbonyl oxygen, which is still there, would also gain an H. This would transform the aldehyde group into a CH2OH group. So we're gonna have CH2OH now, and the rest of the structure would stay the same. So, we'd still have this carbon connected to OHH, and the same with the others. And then CH2OH here on the bottom. Remember, when it comes to naming the common naming of sugar alcohols, we change the 'ose' ending to 'itol.' So, this would be called D-ribitol. This would be the common name of this sugar alcohol.
What is the common name of the sugar alcohol produced when D-galactose is reduced?
L-galactose
D-galactitol
D-galactaric acid
L-galactitol
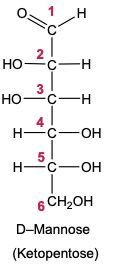
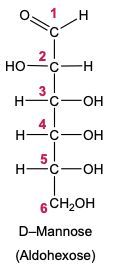
Draw the Fischer projection for the reduction product of D-mannose, the C-2 epimer of glucose. What is the structure and common name of the sugar alcohol produced?
Do you want more practice?
Your GOB Chemistry tutor
- Reduction of d-fructose with a reducing agent yields a mixture of d-sorbitol along with a second, isomeric pro...
- Treatment of d-glucose with a reducing agent yields sorbitol, a substance used as a sugar substitute by people...
- Use the Fischer projection for d-gulose in problem 13.69 to answer each of the following: (13.3, 13.5)a. Draw ...