When it comes to monosaccharides, we say they can exist as either a D enantiomer or an L enantiomer. D versus L stereochemistry is determined by the penultimate carbon. When we say penultimate here, we're talking about the last chiral carbon. Here we have D ribulose versus L ribulose. So how are we able to determine which one is which? We're going to say for the D enantiomer, the penultimate OH, so again, we're talking about the last chiral carbon, which is this one that's circled. If the OH is on the right side, then it's a D sugar. Enantiomer. Here the penultimate OH, again, we're talking about the last chiral center, the one on the very bottom. If it's on the left side, then it's an L sugar. So left L, sugar, L sugar. So again, we're looking at the last chiral center, looking to see what side the OH is on. Is it on the right side making it a D sugar? Is it on the left side making it an L sugar? And we're going to say what's important here is that when we're talking about carbohydrates, at least from a chemistry aspect, we're going to say most carbohydrates found in nature are going to be D sugars. So D sugar is the predominant sugar form that we're going to look at. Alright. So just keep this in mind, when we're talking about D versus L stereochemistry, it's really looking at the last chiral center and determining what side is the OH on. Is it on the left side or is it on the right side?
D vs L Enantiomers - Online Tutor, Practice Problems & Exam Prep
D vs L Enantiomers Concept 1
Video transcript
D vs L Enantiomers Example 1
Video transcript
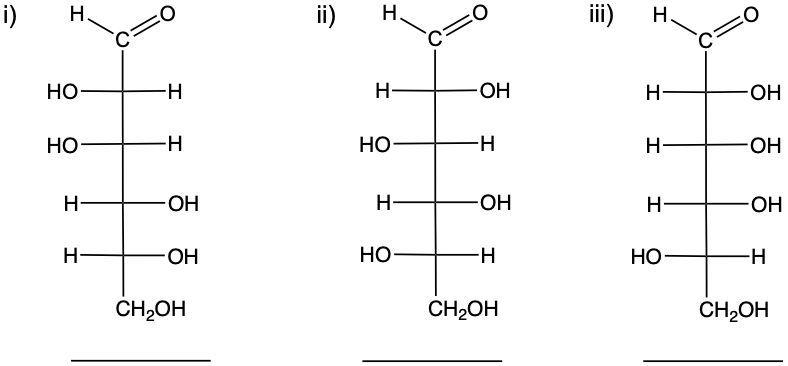
In this example question, it states, "Identify each monosaccharide as D or L enantiomer." So, remember to determine D or L stereochemistry, we look at the penultimate carbon or the last chiral carbon. The last chiral carbon will be this one and this one. If the OH group is on the right side, that makes it a D sugar. So this would be a D enantiomer. And then, if the OH is on the left side (left L), then it's an L enantiomer. So, we would say here that for structure A represents a D enantiomer since the OH is on the right side on the last chiral center, and then we'd say B is an L enantiomer since the OH is on the left side of the last chiral center. Alright. So, that's how we approach this particular example question.
D vs L Enantiomers Example 2
Video transcript
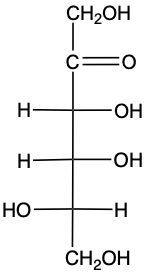
In this example question, it says to draw a C4 epimer of D-fructose. First, we will number this monosaccharide. We start numbering from the end closest to our carbonyl carbon, beginning up here: 1, 2, 3, 4, 5, and 6. The question specifies to design a C4 epimer. An epimer is a type of diastereomer where two structures are similar at all their chiral centers except for one place. Here, that difference is indicated at carbon number 4 (C4). To draw the C4 epimer of fructose, we will ensure that the CH2OH and the carbonyl carbon remain unchanged. We have 1, 2, 3, 4, 5, and 6 in the chain; C3 remains the same. The difference occurs at carbon number 4; we'll flip the orientation so that the OH, which was originally on the right side, is now on the left, and the H is switched accordingly. C5 remains unaffected. This structure now represents the C4 epimer of fructose. We have altered only the chiral center at carbon number 4, which is indicative of a C4 epimer.
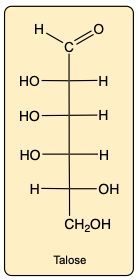
Label each as D-enantiomer, L-enantiomer, epimer or neither of Talose.
D-enantiomer, epimer, L-enantiomer
epimer, neither, L-enantiomer
epimer, L-enantiomer, D-enantiomer
neither, epimer, D-enantiomer
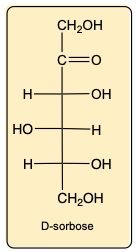
Identify the given molecule as diastereomer or epimer of D-sorbose.
Diastereomer
Epimer
Do you want more practice?
Your GOB Chemistry tutor
- Draw the enantiomer of the following monosaccharides, and in each pair identify the d sugar and the l sugar.a....
- Identify each of the following as D or L:a. <IMAGE>
- Identify each of the following as the D or L enantiomer:a. <IMAGE>
- Draw the Fischer projection for the other enantiomer of a to b in problem 13.21.
- Draw the Fischer projection for the other enantiomer of c to d in problem 13.21.
- Draw the Fischer projections for D-glucose and L-glucose.
- The sugar d-gulose is a sweet-tasting syrup. (13.3, 13.4)<IMAGE>a. Draw the Fischer projection for l-gul...