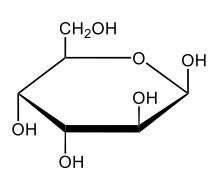
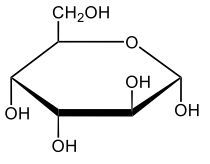
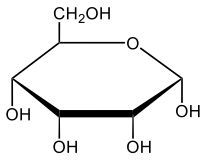
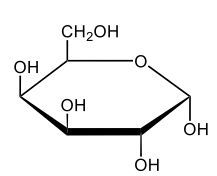
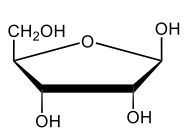
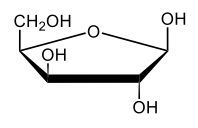
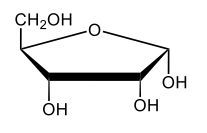
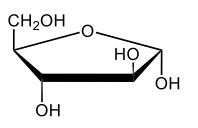
Here we're going to say that monosaccharides exist as cyclic hemiacetals in aqueous solutions. Now cyclization takes place when the penultimate alcohol reacts with the C1 aldehyde group. If we take a look here in the center, we have the acyclic monosaccharide in the form of D-glucose. And we're talking about the penultimate alcohol, so we're talking about this alcohol here. This alcohol is what's going to help to make this within our structure. Now from this acyclic form, we're able to create two possibilities. One where the OH is pointed down when it comes to this hemiacetyl carbon, and one where it's pointing up. But how do we know what they are called? Well, this is where the term anomers comes into play. Anomers, these are just epimers produced by cyclization of monosaccharides. Remember, an epimer means that we have the same configuration at all chiral centers except for one place. If we were to look, all the chiral centers are the same everywhere except for carbon number 1, because that OH could either point down or point up. That gives us 2 epimers or anomers of each other. Here we're gonna say alpha anomer. This is where the OH group and the CH2OH group are on opposite sides of each other. If we can see, the OH group is pointed down here, but the CH2OH group is pointed up. They're opposites of each other, so this would be alpha D-glucose. And then beta anomer, this is where our anomeric OH and C6 CH2OH are on the same side. This will be beta. Now, remember carbon number 1, we can call it the hemiacetyl carbon, but we can also call it the anomeric carbon because it can give us 2 possibilities. The OH could orient itself down, giving us alpha, or could orient itself up, giving us beta. These would be epimers or anomers of each other. Alright. So just remember, we're going from the noncyclic form of glucose, and once we put it in an aqueous environment, it's gonna coil up to form either this ring or this ring as its dominant forms. So keep that in mind when we're talking about different types of cyclic hemiacetals that originate from an acyclic monosaccharide.
Cyclic Structures of Monosaccharides - Online Tutor, Practice Problems & Exam Prep
Cyclic Structures of Monosaccharides Concept 1
Video transcript
Cyclic Structures of Monosaccharides Example 1
Video transcript
Here it says draw on Haworth projection for beta-D-galactose. Alright. So step 1 is we're going to number the Fischer projection. So 1, 2, 3, 4, 5, and 6. And rotate it clockwise to turn it on its side. So now it's 123456. Notice that we're starting out with D-galactose. We don't get beta until it's in its cyclic form. Step 2 now is we're going to curl the CH2OH group clockwise, keeping the carbonyl group in the far right corner. So here we're gonna kind of like bend this structure, it's linear right now, we're gonna bend it a little bit, curve upon itself, so that my CH2OH group and my carbonyl group are in similar positions, similar area with each other. Now, this is gonna be a little bit tricky. We're going to rotate C5, so carbon number 5 here. So, CH2OH faces up, bringing OH group close to the carbonyl group. Alright. So basically, we're gonna have this OH and this CH2OH moving. So this COH is gonna move to where CH2OH is, and then C5H2OH is gonna move here. So that we get now, CH2OH up here, and then the OH rotated over here. And here, we're gonna close the ring to form the cyclic hemiacetal and assign alpha or beta to the anomeric OH group. Remember, the anomeric group is carbon number 1. It is the carbon that was the carbonyl group here. Alright. So basically, this OH and this Carbon are gonna form a bond with each other, this bond here. Oxygen ideally wants to only make 2 bonds, so it's gonna lose its H. And we're gonna say that this double bonded O is no longer gonna be double bonded because carbon can't make 5 bonds. It's gonna transform into an OH group. We said here beta, which means that the OH group has to be on the same side as a CH2OH group. So OH would be up here, oriented up here. And this H is still around and it'd have to be down here. So this here will represent our beta-D-galactose structure.
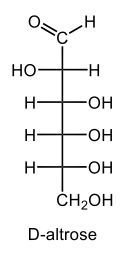
Draw a Haworth projection for α-D-altrose.
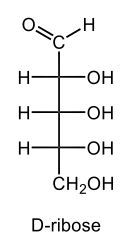
D-ribose is an aldopentose sugar that is found in the DNA. It commonly exists as a five-membered β anomer. Draw D‑ribose in its cyclic hemiacetal form.
Do you want more practice?
Your GOB Chemistry tutor
- In the monosaccharide hemiacetal shown below number all the carbon atoms, identify the anomeric carbon atom, a...
- The cyclic structure of d-idose, an aldohexose, is shown in the margin. Convert this to the straight-chain Fis...
- Identify the following as diastereomers, enantiomers, and/or anomers. (a) ß-d-fructose and ß-d-fructose (b) d-...
- d-Talose, a constituent of certain antibiotics, has the open-chain structure shown next. Draw d-talose in its ...
- l-Fucose is one of the naturally occurring l monosaccharides. It is present in the short chains of monosacchar...
- Draw the structure of the α and ß anomers that result from the reaction of methanol and ribose. Are these comp...
- In its open-chain form, d-mannose, an aldohexose found in orange peels, has the structure shown here. Coil man...
- d-Fructose can form a six-membered cyclic hemiacetal as well as the more prevalent five-membered cyclic form. ...
- Draw the Haworth structures for α- and ß-D-glucose.
- Identify each of the following as the α or ß isomer:a. <IMAGE>
- The sugar d-gulose is a sweet-tasting syrup. (13.3, 13.4)<IMAGE>b. Draw the Haworth structures for α− an...