Now, when it comes to enzyme inhibition, we're going to say that inhibition itself decreases catalyzed reaction rates. And we're going to say that the inhibitor itself is a small molecule or ion that binds to the enzyme and interferes with its activity. Now the characteristics of inhibition, we can talk about it in terms of competitive versus noncompetitive and reversible versus irreversible. Now in competitive inhibition, we're going to say the inhibitor binds to the active site of the enzyme. It's going to block that active site from the substrate, so it can't come in. Noncompetitive inhibition means that the inhibitor binds to a non-active site, so like an allosteric site. And then we're going to say reversible. Well, reversible means that the enzyme can regain or enzyme regains its activity or can regain its activity. Irreversible means that we have a permanent loss of enzyme activity. So these are the different types of inhibitions that overall will hurt or decrease in enzymes' effectiveness and activity.
Enzyme Inhibition - Online Tutor, Practice Problems & Exam Prep
Enzyme Inhibition Concept 1
Video transcript
Enzyme Inhibition Concept 2
Video transcript
So in this video, we're going to take a look at the different types of inhibition: Competitive versus noncompetitive, reversible versus irreversible. So if we take a look at the first one, we're going to say we have reversible competitive inhibition. Here we have our substrate, and within the active site of our enzyme, we have the inhibitor in place. Now, here we're going to say that the inhibitor is similar in shape and size to the substrate. So it can take up the active site instead of the substrate. A great example of this is Ibuprofen. Now, what's the interaction here? Well, here this would be a non-covalent interaction between the inhibitor and the enzyme at the active site. And what is the effect? Well, the effect we can see is that it's taking up that active site position, so it's blocking it from the substrate. And how do we reverse this effect? How could we get that inhibitor to not get in the way of our substrate? Well, increasing the concentration of our substrate would help to lower the effect of this inhibitor.
Now, reversible noncompetitive. Here, we have our active site, we have our substrate, and then down here we have our inhibitor. It attached somewhere else that is not the active site. So here we're going to say that when it comes to the inhibitor, it does not resemble the substrate's shape because it's not binding to the active site where the substrate would go. A great example of this are heavy metals. Now here this is also non-covalent in terms of interaction, but now it's at the non-active site. Here, we're going to say it causes a shape change in the enzyme and in the active site. But by the enzyme, the inhibitor attaching itself to this part down here, which is not the active site, it's actually going to cause a shift or change in the shape of this active site. It changes shape so the substrate can no longer attach effectively to the enzyme. Here, we're going to say, how could we reverse this effect? Well, we have to use special types of agents. They would have to bind to the inhibitor so that they cannot bind to the non-active site and change the active site of the enzyme.
Finally, we have irreversible. Here we have our substrate, we have our enzyme, and the inhibitor has attached to the active site. Here we would say that when it comes to the inhibitor, it does not resemble the substrate's shape. In this case, we could talk about poisons and different types of venoms belonging to this type of inhibition. Now, here we're going to say we have a covalent interaction with our group within the active site. And we're going to say here that the effect is it blocks the active site. The inhibitor is attaching itself to that active site. And here the inhibition is permanent. So this can do some long-lasting damage and effects in terms of irreversible inhibition. So we've talked about the different types of inhibition, remember, competitive versus noncompetitive, reversible versus irreversible.
Enzyme Inhibition Example 1
Video transcript
Here in this example question, it says, match each type of inhibition with the correct statement. So a is reversible competitive, b is reversible noncompetitive, and c is irreversible. The first one says, inhibitor decreases enzyme activity by binding at a site other than the active site. Well, remember, in our different types of inhibition that we covered, we'd say that the reversible noncompetitive is the only one that has the inhibitor binding to a site that is not the active site. So first, it's going to be B. Next, the formation of strong covalent bond leads to permanent loss of enzyme activity. So, remember, when it comes to irreversible inhibition, it's the covalent of the R groups that are binding to the active site. Also, it says permanent. Remember, the effect here is permanent when it comes to irreversible. So this would be C. Next, the substrate is unable to bind with the active site due to a change in enzyme structure. Alright. So for this one, they're talking about the active site changing form. This happens when the inhibitor attaches to the nonactive site. It causes what we call a conformational change in our active site. The shape changes so the substrate can no longer effectively attach to it. This happens with reversible noncompetitive inhibition. Next, an increase in the concentration of substrate increases the reaction rate. We said that the reversible competitive inhibition, its effects can be reversed if we increase the amount of substrate. This is going to put the inhibitor at a disadvantage where it won't be able to effectively bind to the active site. So here, this would be A. So we'd say that the statements would be B, C, B, and A for this particular example question.
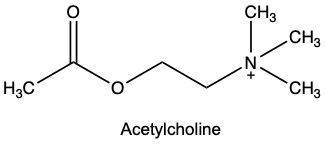
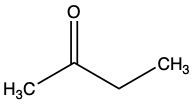
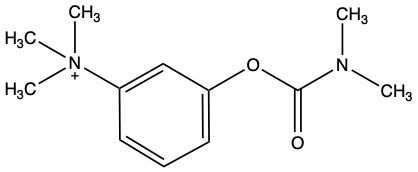
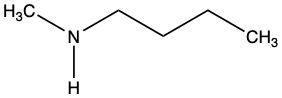
Which of the following inhibitors could be a reversible competitive inhibitor for acetylcholinesterase if its substrate is acetylcholine?
Do you want more practice?
Your GOB Chemistry tutor
- How can you distinguish between a competitive inhibitor and an uncompetitive inhibitor experimentally?
- Why are irreversible enzyme inhibitors referred to as poisons?
- The text discusses three forms of enzyme inhibition: uncompetitive inhibition, competitive inhibition, and irr...
- What kind of inhibition (uncompetitive, competitive, or irreversible) is present in each of the following:a. P...
- What kind of inhibition (uncompetitive, competitive, or irreversible) is present in each of the following:c. T...
- One mechanism by which lead exerts its poisonous effect on enzymes can be stopped by chelation therapy with ED...
- What kind of reaction product might be a competitive inhibitor for the enzyme that catalyzes its formation?
- Explain how the following changes affect the rate of an enzyme-catalyzed reaction in the presence of an uncomp...
- Explain how the following changes affect the rate of an enzyme-catalyzed reaction in the presence of an uncomp...
- What type of enzyme regulation occurs in the following situations?d. Conversion of isocitrate to α-ketoglutara...
- Indicate whether each of the following statements describes primary, secondary, tertiary, or quaternary protei...
- Indicate whether each of the following describes a competitive or a noncompetitive enzyme inhibitor:d. The str...
- Methanol and ethanol are oxidized by alcohol dehydrogenase. In methanol poisoning, ethanol is given intravenou...
- In humans, the antibiotic amoxicillin (a type of penicillin) is used to treat certain bacterial infections.a. ...
- Oxaloacetate is an inhibitor of succinate dehydrogenase. <IMAGE> a. Would you expect oxaloacetate to be ...
- In humans, the antibiotic amoxicillin (a type of penicillin) is used to treat certain bacterial infections.c. ...
- Ethylene glycol (HO—CH₂—CH₂—OH) is a major component of antifreeze. If ingested, it is first converted to HOOC...