Now recall that amides consist of a carbonyl group connected to a nitrogen, which could have its origins from an amine group. Amides can be classified as either being primary, secondary, or tertiary. Here we have an example of an amide: at the far right, we have our carbonyl connected to our nitrogen group. Whether you're primary, secondary, or tertiary is based on how many carbons are directly attached to the nitrogen. In this first example, there is only one carbon directly attached to the nitrogen, so this is a primary amide. For the next one, there are two carbons directly attached to the nitrogen, so this is secondary. And then here there are three, so this is tertiary. That's how we look at the substitution around amides: it's the number of carbons directly attached to the nitrogen.
Naming Amides - Online Tutor, Practice Problems & Exam Prep
Intro to Amides Concept 1
Video transcript
Intro to Amides Example 1
Video transcript
So here we have to classify each of the given compounds as primary, secondary, or tertiary amides, or neither. Now here, if we take a look, this is an amide because we have a carbonyl connected to a nitrogen directly, and there's only one carbon directly connected to that nitrogen, so it is a primary amide. For the next one, there is no carbonyl present, so it's just sp3 carbons connected to a nitrogen. So this is actually an amine, but we're not asking about amines, so we'll put neither. For the next one, we have our carbonyl directly connected to a nitrogen. So this is an amide and there are 1, 2, 3 carbons directly attached to it. So this is a tertiary amide. Then finally, for this last one, it is also an amide, and it's directly connected to 1, 2 carbons in terms of the nitrogen. So this would be a secondary amide. And that's how we classify each of the given compounds.
IUPAC Rules for Naming Amides Concept 2
Video transcript
So when it comes to the IUPAC naming of amides, we're going to see the carbon chain with the carbonyl group is named as though it was a carboxylic acid. Remember, carboxylic acids end with oic acid. So what we do here is we modify the ending from oic acid to just amide. And our naming convention will be our substituents that are connected to our parent. And we're modifying the ending from again oic acid to amide. So this is the approach we're going to take when it comes to IUPAC naming of different types of amides that we'll see.
IUPAC Rules for Naming Amides Example 2
Video transcript
Provide a systematic name for the following amide. Now, here's step 1: we have to identify the alkyl group or groups connected to the nitrogen atom. So, we're talking about this right here. That is a one-carbon alkyl group, so it's a methyl group. Next, we're going to name the alkyl groups as substituents, which means they're going to go at the beginning of the name. And we're going to say here, n, and this is important, it has to be italicized. This will represent an n substituent, and it's written before the alkyl name to indicate it's connected to the nitrogen atom. Now, when more than one identical substituent, we're going to use the prefix di. Step 3 is we identify the carbon chain connected to the carbonyl group, so C double bond O. Step 4 is we figure out the length of the carbon chain starting from the carbonyl group, and we're going to say the carbonyl group as carbon number 1 is implied. From that point, we use the same rules that we learned in terms of naming esters to name this particular amide. Right. So, if we take a look here, so we have a methyl group here, and it's an n-methyl since it's connected to the nitrogen. Remember, it has to be written in italics. And then we're going to say here, this chain with the carbonyl is 1, 2, 3, 4, and 5. So that's 5 carbons there. And then we have this ethyl group over here that is also another substituent. It's located on carbon number 3. So, in alphabetical order, 'e' comes before 'm', so 3-ethyl-n-methyl. It's a 5 carbon chain, so that's pentane, but here we're going to change the e-ending of pentane to amide, so it's pentanamide. So, this will be the name of this particular amide.
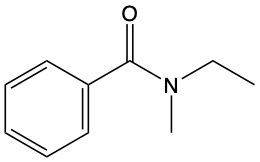
Give IUPAC name for the following amide.
ethylmethylbenzoicamide
N-ethyl-N-methylbenzamide
ethylmethylbenzamide
N-methyl-N-ethylbenzoic amide
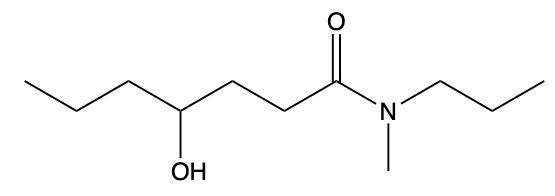
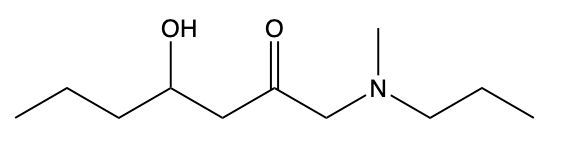
Draw structure from provided IUPAC name: 4-hydroxy-N-methyl-N-propylheptanamide.
Common Naming: Amides Concept 3
Video transcript
So when it comes to the common naming of an amide, we're going to say they follow the same rules as IUPAC, except the parent chain consists of the common name prefixes. Now, the modification of the ending is still going from oic acid to amide. So, if we take a look at this example, it says to provide a common name for the following amide. So if we take a look, we’d say that we have a methyl and then 1, 2, 3, 4, a butyl attached to the nitrogen. Those are our Nsubstituents. Alphabetically, b comes before m. So we’d have N-butylN-methyl, and then the chain with the carbonyl has 1, 2, 3 carbons. Since this is a common name, the prefix will be propion, and then it is an amide, so we just add amide to the end. So this would be the name, the common name, of this particular amide structure.
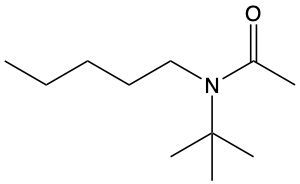
Assign a common name to the following.
N-pentyl-N-tert-butylacetamide
N-tert-butyl-N-pentylethanamide
N-tert-butyl-N-pentylacetamide
N-hexyl-N-tert-butylethanamide
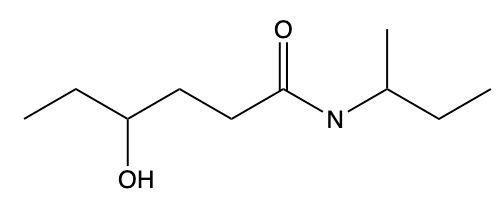
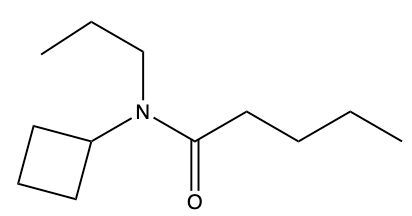
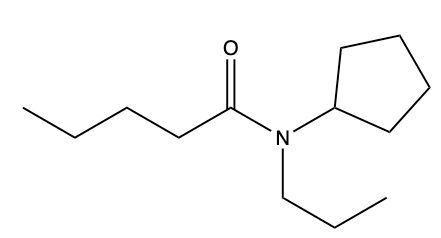
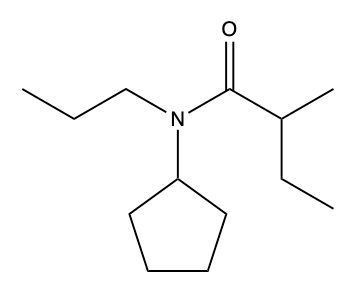
Draw a structure from the following name: N-cyclopentyl-N-propylvaleramide.
Do you want more practice?
Your GOB Chemistry tutor
- What are the names of the following compounds?c. <IMAGE>
- Identify the following molecules as an ester, a carboxylic acid, or an amide, and write both the condensed and...
- Give systematic names for the following structures and structures for the names:c. N-Ethyl-N-methylbenzamide
- Give systematic names for the following structures and structures for the names:a. 3-Methylpentanamide
- Give systematic names for the following structures and structures for the names:b. N-Phenylacetamide
- Write the IUPAC and common names, if any, for each of the following amides:c. <IMAGE>
- Draw the condensed structural formula for each of the following amides:b. 2-methylpentanamide
- Draw the condensed structural formula for each of the following amides:a. heptanamide
- Draw the condensed structural formula for each of the following amides:c. 3-methylbutyramide
- There are four amide isomers with the molecular formula C3H7NO. Draw their condensed structural formulas and w...
- Write the IUPAC name for each of the following: (14.6)b . <IMAGE>
- Write the IUPAC and common name, if any, for each of the following carboxylic acids:b. <IMAGE>