Hey, everyone. So in this video, we're going to take a look at acid catalyzed hydrolysis of esters. Now under this reaction, an ester is hydrolyzed into a carboxylic acid and an alcohol in an acidic medium. Now when we say hydrolyzed and we say a hydrolysis reaction, well, this just means a reaction that breaks out a molecule through a reaction with water. And the basic idea of this reaction is that if we take a look here at this general reaction here, we have our ester bond here, this blue part here, and water with the use of an acid catalyst in the form of H+, it's going to be used to cut this ester bond. As a result of this, the carbonyl carbon gains an OH. So here our carbonyl carbon gains an OH and it becomes a carboxylic acid, and then the oxygen which was part of the ester linkage will gain 1 hydrogen atom. This helps to create it into an alcohol. So the basic idea of this reaction is that we have an ester and through the use of water and an acid catalyst, we help to create a carboxylic acid and an alcohol as our two products.
16. Carboxylic Acids and Their Derivatives
Ester Reactions: Acid-Catalyzed Hydrolysis
16. Carboxylic Acids and Their Derivatives
Ester Reactions: Acid-Catalyzed Hydrolysis - Online Tutor, Practice Problems & Exam Prep
1
concept
Acid-Catalyzed Ester Hydrolysis Concept 1
Video duration:
1mPlay a video:
Video transcript
2
example
Acid-Catalyzed Ester Hydrolysis Example 1
Video duration:
47sPlay a video:
Video transcript
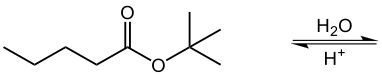
Draw skeletal formulas for the products of the following reaction. So here we have our ester, and we're using water with h+ as a catalyst. Now remember, the premise of this reaction is we're going to cut the ester bond here. As a result of this, the carbonyl carbon here will gain an OH group to become a carboxylic acid, and then we're going to say that oxygen here will gain a hydrogen and create an alcohol. So in this acid-catalyzed hydrolysis, we have our ester being transformed into a carboxylic acid and an alcohol as our two products.
3
Problem
ProblemWrite a common name for the carboxylic acid produced in the following reaction.
A
Oxalic acid
B
Valeric acid
C
Butyric acid
D
Malonic acid
Do you want more practice?
We have more practice problems on Ester Reactions: Acid-Catalyzed Hydrolysis
Your GOB Chemistry tutor
Additional resources for Ester Reactions: Acid-Catalyzed Hydrolysis
PRACTICE PROBLEMS AND ACTIVITIES (7)
- Draw the products you would obtain from acid-catalyzed hydrolysis of the following esters.a. Isopropyl benzoat...
- Draw the products you would obtain from acid-catalyzed hydrolysis of the following esters.b. <IMAGE>
- Draw the condensed structural or line-angle formulas for the products of the following:b. <IMAGE>
- Ethyl octanoate is a flavor component of mangoes.c. Write the balanced chemical equation for the acid hydrolys...
- Draw the condensed structural or line-angle formulas for the products from the acid- or base-catalyzed hydroly...
- Draw the condensed structural or line-angle formulas for the products from the acid- or base-catalyzed hydroly...
- The ester methyl butanoate has the odor and flavor of strawberries. (14.3, 14.4)c Write the balanced chemical...