In this video, we're going to take a look at carboxylic acid reactions. Now here we do this by examining acid-base reactions. We're going to say, under this type of reaction, a base reacts with a carboxylic acid, which represents a weak acid. If we take a look here, we have a carboxylic acid in the form of ethanoic acid. Here we're going to use some type of base, which is going to remove an H+ from the carboxylic acid, and in doing this creates its conjugate base form, which is a carboxylate anion. Now, here the carboxylate anion, it's ending differently. In its carboxylic acid form it's oic acid. In its carboxylate anion form, you change the oic acid to oate. So ethanoic acid becomes ethanoate. Right? So just remember, when we're talking about a carboxylic acid reaction, we're basically saying the carboxylic acid is an acid, we can tell from its name, and it's reacting with some type of base which removes an H+ from it, thereby creating a carboxylate anion product.
Carboxylic Acid Reactions - Online Tutor, Practice Problems & Exam Prep
Acid-Base Reactions Concept 1
Video transcript
Carboxylic Acid Reactions Example 1
Video transcript
Predict the products formed from the following reaction between propanoic acid and ammonia. Alright. So here, this is our acid, ammonia, which is a neutral nitrogen compound we know is our weak base. Now, since both of these species are weak, we know that we have reversible arrows to show that an equilibrium has been established. If you don't remember this, make sure you go back and take a look at my videos, where we talk about reactions and equilibrium between acids and bases.
So we know following the Bronsted and Lowry definition of acids and bases, that the carboxylic acid gives away an H+ to the ammonia, it becomes a carboxylate anion in the process. The ammonia gains an H+ to become the ammonium ion, so it becomes NH4+. So although we're talking about an organic molecule in terms of carboxylic acid, this is kind of familiar because it follows our Bronsted-Lowry definitions of acids and bases. The acid donates an H+, the base accepts the H+. And in that way, we make our two ions as products.
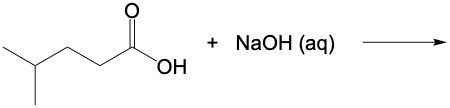
Name the carboxylate anion formed in the following reaction.
2-methylpentanoate
4-methylhexanoate
4-methylpentanoate
2-methylhexanoate
Do you want more practice?
Your GOB Chemistry tutor
- Write the products of the following reactions:b. 2, 2-Dimethylpentanoic acid + KOH → ?
- Write the equation for the ionization of hexanoic acid in water at pH 7.4. (Hint: See Section 17.2.)
- Suppose you have a sample of benzoic acid dissolved in water.b. Now assume that aqueous NaOH is added to the b...
- Suppose you have a sample of benzoic acid dissolved in water.c. Finally, assume that aqueous HCl is added to t...
- A simple polyamide can be made from ethylenediamine and oxalic acid (Table 17.1). Draw the polymer formed when...
- Write the balanced chemical equation for the dissociation of each of the following carboxylic acids in water:a...
- Write the balanced chemical equation for the dissociation of each of the following carboxylic acids in water:b...
- Identify the compound in each group that is most soluble in water. Explain.b. pentane, 1-hexanol, propanoic ac...
- Identify the compound in each group that is most soluble in water. Explain.b. ethanoic acid (acetic acid), hex...
- Draw the condensed structural or line-angle formulas for the products of the following: (14.2, 14.3, 14.6) ...
- Write the balanced chemical equation for the reaction of each of the following carboxylic acids with NaOH:c. b...
- Write the balanced chemical equation for the reaction of each of the following carboxylic acids with KOH:b. 2-...