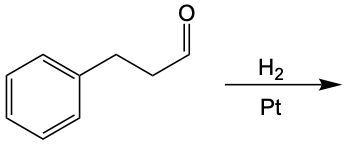
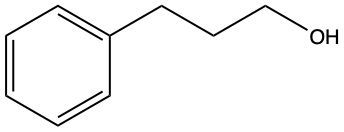
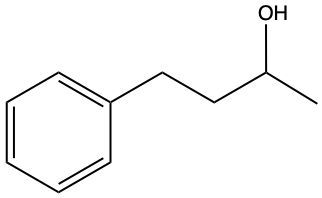
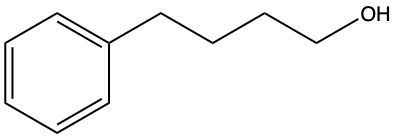
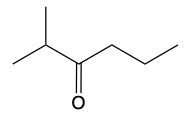
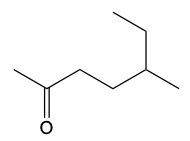
Now, when it comes to the reduction of aldehydes and ketones, recall that reduction uses a reducing agent to add as many carbon-hydrogen bonds as possible without breaking any carbon-carbon bonds. So that's the whole idea of reduction. If we take a look here, we're only paying attention to what's within this shaded box. That's what's really the most important. But stuff lies outside that box, so let's take a look. So if we're starting here over here on the right, we can say that the most oxidized form of carbon exists as carbon dioxide. Carbon is making its maximum 4 bonds, all of them being to oxygen. Once I start the reduction process, we're going to reduce it some, and we're going to start to add as many carbon-hydrogen bonds as possible. Carbon can only go up to 4 bonds. So to do that, we have to break one of these carbon-oxygen bonds. So that's where this H comes in. Carbon needs to keep making 4 bonds, so that's where this H comes in. Oxygen also needs to make its ideal number of bonds, which is 2. So we add a hydrogen to it as well. If we keep going, we enter the shaded region here. So here we keep adding carbon-hydrogen bonds, so we have to get rid of this bond here, and that's how we wind up with this aldehyde. This aldehyde can undergo further reduction, so we're going to add more carbon-hydrogen bonds to it. Meaning we have to break this carbon-oxygen bond here. So here goes the H that I've added to the carbon, oxygen still wants to keep making 2 bonds, so here it would have its hydrogen added to it as well. So we can see here that reducing an aldehyde has created an alcohol. If we wanted, we could continue further with strong reduction to help create an alkane at the end. The alkane being the last stop in terms of reduction process. But what we need to take from this is that we can reduce an aldehyde into an alcohol. And even if we had a ketone, we could do the same. Because if we reduced that, we want to add as many carbon-hydrogen bonds as we want. So we'd again remove one of these carbon-oxygen bonds. So we'd have our carbon with its 2 carbons, 1 on each side, its newly gained hydrogen, and then the oxygen would also gain a hydrogen. So we can see here that we can reduce both aldehydes and ketones to produce alcohols. Okay. So that's the whole basis when it comes to reducing aldehydes and ketones. Doing so changes them both into alcohols.
Reduction of Aldehydes and Ketones - Online Tutor, Practice Problems & Exam Prep
Reduction Reactions Concept 1
Video transcript
Reduction of Aldehydes and Ketones Example 1
Video transcript
Which of the following compounds could not be reduced? So if we take a look at our options, we have 2,2-dimethylpentane, 2-methyl-1-pentanal, 3-ethyl-2-heptanone, and then 4-bromoheptanoic acid. Now we don't need to draw all these. The names themselves tell us the identities or functional groups. Pentane, this is an alkane. Al means this is an aldehyde. One means that this is a ketone. And then oic acid, pentanoic acid that tells us this is a carboxylic acid. Alright. So remember the whole line in terms of reduction. We start out at carbon dioxide as the most oxidized form of carbon. When we reduce that, we change it into a carboxylic acid. It says what cannot be reduced? Well, we know that a carboxylic acid could be reduced if we wanted to, we could reduce it to an aldehyde. And we know that an aldehyde can be reduced to an alcohol. So this is out, and this is out. We also know that a ketone can be reduced to an alcohol as well just like an aldehyde. The answer here is option A, an alkane. Remember we said that the alkane is the end stop of our reduction line, right? Once we get to that point, we've added as many hydrogens as we can to our carbon. So there's nothing else we can do. Remember we cannot break carbon-carbon bonds. So even if that carbon is connected to another carbon, there's nothing we can do to sever that bond to add another hydrogen. So an alkane represents the most reduced form, therefore it cannot undergo reduction, and therefore option A is the answer.
Reduction of Aldehydes and Ketones Concept 2
Video transcript
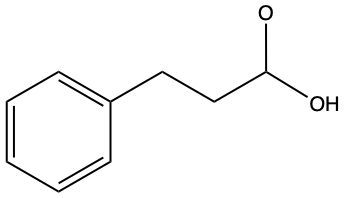
Now, we can say here that aldehydes and ketones are reduced to primary and secondary alcohols respectively. The reducing agent within these processes is H2. And the catalyst that helps facilitate this reduction, we can have them as metals which are Ni (nickel), platinum, or palladium. Now the whole process, the whole result of this reduction is that the carbonyl oxygen gains an h and the carbonyl carbon also gains an h. If we take a look here, we start out with an aldehyde. We're reducing it with H2 and nickel. Remember reduction means we have to try to make as many carbon-hydrogen bonds as we can without breaking any carbon-carbon bonds. To do this, we'd have to break one of the carbon-oxygen bonds here. And when we break that bond, carbon still needs to make its 4 bonds and oxygen would ideally want to still make its 2. So, this H2 basically gives itself up: one h goes to the carbon, the other h goes to the oxygen. As a result of this, we see that we made a primary alcohol.
A ketone, well, a ketone follows the same process. We're going to say we use H2. And remember, we could use nickel, platinum, or palladium as our metal catalyst. And again, we're trying to get rid of one of these carbon-oxygen bonds. To make up for that loss, carbon's going to bond to a hydrogen and oxygen is going to bind to a hydrogen. What we make now is a secondary alcohol. So just remember, when it comes to reduction, both aldehydes and ketones can be reduced. Aldehydes typically create primary alcohols. Ketones primarily make secondary alcohols. Okay. So, they're going to make secondary alcohols once they undergo reduction. Alright. So just keep that in mind.
Reduction of Aldehydes and Ketones Example 2
Video transcript
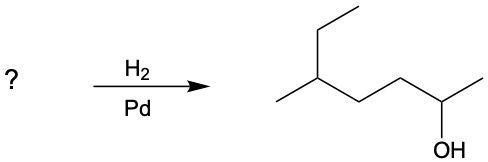
Determine the alcohol product formed in the following reaction. So here we have a ketone, and we're using our reducing agent of H2 with our metal catalyst, nickel. Remember, ketones get reduced to secondary alcohols. What's going to happen here is we're going to cut one of these carbon-oxygen bonds, and to maintain 4 bonds for carbon and 2 bonds for oxygen, each one of them gains a hydrogen. So what we'll have here is we'll have CH3CH2 still. This is still connected to this carbon, which is still connected to this oxygen. And then we have CH2CH3 again. So this H2 here, one of the H's will go to carbon and the other H will go to oxygen. We've just created a secondary alcohol by reducing the ketone reactant.
Determine the alcohol product formed in the following reaction.
Determine which reactant should be used to produce the following alcohol.
Do you want more practice?
Your GOB Chemistry tutor
- The carbonyl group can be reduced by addition of a hydride ion (H⁻) and (H⁺) a proton . Removal of H⁻ and H⁺ f...
- The carbonyl group can be reduced by addition of a hydride ion (H⁻) and (H⁺) a proton . Removal of H⁻ and H⁺ f...
- Write the structures of the hemiacetal or hemiketal that result from reactions (a) and (b). Write the structur...
- What ketones or aldehydes might be reduced to yield the following alcohols?a. <IMAGE>b. <IMAGE>c. ...
- Draw the structures of the products formed when the following compounds react with a reducing agent.<IMAGE&...
- Can the alcohol (CH3)3COH be formed by the reduction of an aldehyde or ketone? Why or why not?
- Indicate which compounds contain aldehyde or ketone carbonyl groups.<IMAGE>
- Indicate which compounds contain aldehyde or ketone carbonyl groups. CH3CH2―O―CH2―CHO
- Indicate which compounds contain aldehyde or ketone carbonyl groups. <IMAGE>
- Draw the condensed structural formula for the alcohol formed when each of the following is reduced by hydrogen...
- Draw the condensed structural formula for the alcohol formed when each of the following is reduced by hydrogen...
- Draw the condensed structural formula for the alcohol formed when each of the following is reduced by hydrogen...
- Draw the condensed structural formula for the alcohol formed when each of the following is reduced by hydrogen...
- Draw the condensed structural or line-angle formula for the alcohol produced when hydrogen and a nickel cataly...
- Draw the condensed structural or line-angle formula for the alcohol produced when hydrogen and a nickel cataly...
- Draw the condensed structural formula for the alcohol formed when each of the following is reduced by hydrogen...