Now, what carbon hydrocarbons are the simplest organic compounds composed solely of carbons and hydrogens. Here in this list, we're going to go over different types of hydrocarbons. So, if we start with the first row, we have alkanes. Alkanes are just carbons that are single bonded to one another. Now if we pay attention, remember, each one of these bonds connects two carbons together, carbon must make 4 bonds. If we look at the two carbons in the middle, they each have two hydrogens. Now, focus on either one of these carbons, and you would see that they are connected to 4 surrounding atoms, meaning that their hybridization is sp3. Their generic formula, if we were to look here, we'd have 4 carbons, and you would have 10 hydrogens. And that's because the generic formula for an alkane is CnH2n+2, where n is the number of carbon atoms. So if we said that we need an alkane that has 5 carbons, what is its molecular formula? It'd be C5H12. Now, alkenes is when you have two carbons double bonded to each other; they still need to make 4 bonds, they're making 3, so they each have one hydrogen that's invisible. Notice that the number of hydrogens decreased as we added that pi bond between the carbons. These carbons here are connected to three surrounding atoms, so they are sp2 in terms of hybridization. Each of these double-bonded carbons lost a hydrogen, so we lost two hydrogens overall. So our generic formula has changed to CnH2n. We've dropped the plus 2 because we lost those two hydrogens. Alkynes are triple bonded carbons, so there's one here and here. They're making four bonds right now, so they don't need any extra hydrogens on them. These two sp carbons are sp hybridized because they are connected to two surrounding atoms. And think about it, we just lost another two hydrogens, so the generic formula becomes CnH2n-2. Cycloalkanes are just alkanes that are in rings. So they're all still single bonded. In this ring, we have carbons all around. Each of these carbons has two hydrogens on them, right? So if we think about it, focus on one of them. Let's focus on this one right here. It is connected to four surrounding atoms, so it is sp3 hybridized. Here if we look, we have five carbons for ten hydrogens, so the ratio is there's double the amount of hydrogens to carbon. So that means the formula will be CnH2n. So a Cycloalkane has the same generic formula as an alkene. We have to lose two hydrogens in order to enclose this alkane into a ring. Finally, aromatic refers to a Benzene ring. A Benzene ring is six carbons in a ring with alternating double bonds. Each of these carbons has one hydrogen. So a Benzene has a formula of C6H6. Another way of showing it is like this. This is changing it into more of a skeletal formula. Each of those carbons are double-bonded, they're each connected to three surrounding atoms, so they are sp2 hybridized. And here it's a one-to-one relationship between Carbon and Hydrogen since it's C6H6. So the generic formula is CNHN. Alright. So these encompass all the different types of hydrocarbons that you may encounter when dealing with deeper types of ideas in organic chemistry. But remember, essentially they're all just compounds with carbons and hydrogens. Here some of them have single bonds, some of them have double bonds, some of them have triple bonds or are in rings. Okay. But they're all hydrocarbons at the end of the day.
Intro to Hydrocarbons - Online Tutor, Practice Problems & Exam Prep
Intro to Hydrocarbons Concept 1
Video transcript
Intro to Hydrocarbons Example 1
Video transcript
So in this example question, it says, classify each of the following hydrocarbons as an alkane, alkene, or alkyne. So remember, an alkane is when we have carbons just single bonded to one another. If we take a look at the options, remember every line connects 2 carbons to each other. In this option B, all of them are single bonded to one another, so they're all alkanes. Well, it is an alkane. This is the only alkane that we have. An alkene is when we have carbons double bonded to one another. These 2 carbons here are double bonded to each other, and as long as 2 carbons are double bonded to each other, it is an alkene. These 2 carbons are double bonded to each other, so this is also an alkene. An alkyne is when we have 2 carbons triple bonded to one another. We have a carbon here, and a carbon here. So option C would represent an alkyne. This is how we describe our options A, B, C, and D for the following example question.
Saturated and Unsaturated Hydrocarbons Concept 2
Video transcript
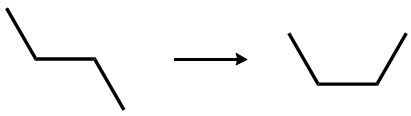
Now with hydrocarbons, we now talk about saturated versus unsaturated. A saturated hydrocarbon means that all the bonds are single bonds. All the carbons connected to each other have single bonds, which allows each carbon to have the maximum possible number of hydrogen atoms. Consider the image here, where the one on the left is a saturated hydrocarbon. Those three carbons have to make four bonds each, and to achieve this, we'd have to add hydrogens to them.
An unsaturated hydrocarbon means that there is at least one double bond or triple bond, implying that we won't be able to have a maximum number of hydrogens. Taking a look at this double bond here, what effect will that have? The carbon on the left is single bonded, so it still has its maximum number of three hydrogens. However, because of that double bond there, remember each carbon needs to make four bonds. This carbon here is already making three bonds, so it only needs one hydrogen. The same applies to this carbon, which also needs one additional hydrogen. What's the difference? Well, these two carbons, the one here had two hydrogens, but with the inclusion of a double bond, now it only has one. This carbon here has three hydrogens, but the inclusion of the double bond means now it has only two. So, adding a Pi bond or a double bond means that we're going to have to forfeit one hydrogen atom per carbon. And if we were to make a triple bond, again we'd need to forfeit one hydrogen atom between those carbons. & Therefore, this illustrates the difference between a saturated hydrocarbon, where there are no double bonds or triple bonds, so you have the maximum of hydrogen atoms on carbons, and an unsaturated hydrocarbon, where now double bonds and triple bonds decrease the total number of H atoms possible.
Saturated and Unsaturated Hydrocarbons Example 2
Video transcript
In this example question, it says, classify the following hydrocarbons as saturated or unsaturated. Remember, a saturated hydrocarbon possesses only single bonds between its carbons. In this image, a and c would represent saturated hydrocarbons. Every line connects carbons together. All these carbons are single bonded to one another, meaning they'll have a maximum number of hydrogen atoms attached to each of them. Unsaturated would be these 2. Unsaturated hydrocarbons have a double bond or a triple bond present. This decreases the maximum number of H atoms that can connect to a potential carbon. Remember, carbon must make 4 bonds. Adding those Pi bonds decreases that number. Alright. So a and c would be saturated, b and d would be unsaturated.
Write the molecular formula for an alkane with 5 C atoms.
C5H12
C5H10
C5H14
C5H8
Write the molecular formula for an alkyne with 4 C atoms.
C4H8
C4H10
C4H6
C4H4
Which of the following molecular formulas might indicate an alkene?
C7H16
C6H12
C5H8
C4H10
Bond Rotation and Spatial Orientation Concept 3
Video transcript
So in our continued discussion of hydrocarbons, we're now going to take a look at bond rotation and spatial orientation. Here we're going to say carbon-carbon bonds in alkanes can rotate freely, and that's because of the presence of a single bond. So here, this is a carbon and this is another carbon, and we rotate around this single bond. Now by rotating that, we're going to keep one side stationary, meaning we're not going to move it. We're going to keep this side still, so it matches up with this side. And we're going to rotate the other part, the right side. When I'm rotating, this red ball is up here, so when I rotate, it'll rotate down to where the blue is. So there it goes right there. And then when I rotate that down, this blue comes to where this part is, so there goes the blue part right there. And then that green thing has to shift up to this, to where the red was, so there's the green part right up here.
How would this look like in terms of a skeletal formula? So again, there's free rotation around the single bond, so we rotate, we keep this side stationary, so it stays where it is. Everything else is spinning. This O would rotate down to where the H is, so that's why it's here. This H would rotate to where this methyl group is, and then this, this CH3 group here would rotate up here.
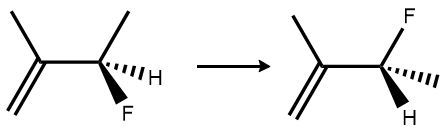
Now, if we have a double bond, which is in alkenes, we cannot rotate. There's no free rotation around a pi bond, a double bond. This leads to two different spatial orientations and therefore two different compounds. These two carbons are double bonded, so we can't rotate. That means that these two blues are locked on the same side, and these two greens are locked on the same side. So it would not be equal to this molecule here, where the blues are opposites of each other and the greens are on opposite sides of each other. If we show this as a skeletal formula, these two H's are locked on the same side, but here they're on opposite sides. There's no free rotation, so these will represent two different compounds.
So just keep in mind when we're dealing with double bonds and also triple bonds, there's no free rotation around that bond. Things are locked where they are.
Bond Rotation and Spatial Orientation Example 3
Video transcript
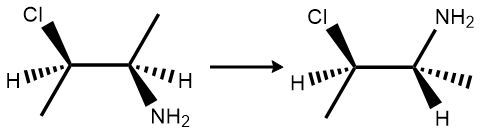
Here it says determine if the two structures below are the same or different compounds. Well, both compounds have 2 carbons double bonded to each other. Remember, there is no free rotation around that double bond, so things are stuck where they are. The one on the left has an H and an F next to each other, but the one on the right has 2 H's instead next to each other. The one on the left has a Br and an H next to each other, but this one here has a Br and an F next to each other. We have different groups next to one another in both molecules or both compounds, and we can't rotate around that double bond so that they're the same. Because of this, they would represent different compounds. So here, our answer would be option b.
Which of the following is not a valid bond rotation?
Do you want more practice?
Your GOB Chemistry tutor
- What is wrong with the following structures?CH₃=CHCH₂CH₂OH
- There are two things wrong with the following structure. What are they? <IMAGE>
- What do the terms saturated and unsaturated mean?
- Draw an example of a saturated four carbon compound and an unsaturated four carbon compound.
- Are the pairs of compounds shown below the same molecule, isomers, or different molecules? <IMAGE>
- Identify the following as alkanes, alkenes, cycloalkenes, or alkynes:d. <IMAGE>
- Identify the following as alkanes, alkenes, cycloalkenes, or alkynes:a. <IMAGE>
- Indicate whether each of the following pairs represent structural isomers or the same molecule:b. <IMAGE>...
- Identify the family of hydrocarbon present in the following:(c) <IMAGE>